Naming Compounds
Ionic
Metal always on the left
Most left on the periodic table goes on the left (positive charge on left)
Writing Chemical Formula
Writing Compound Name
Just write the name of the elements
Last element takes 'ide'
Roman Numeral is written after the metal if the combining capacity is different
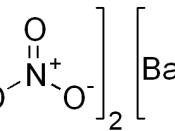
Copper (II) Nitrate
MgS = Magnesium Sulphide
Covalent
Writing Compound Name
Write prefixes for both elements
'mono' is dropped for the 1st element
1 Mono 6 Hexa
2 Di 7 Hepta
3 Tri 8 Octa
4 Tetra 9 Nona
5 Penta 10 Deca
NO2 = Nitrogen Dioxide
CO = Carbon Monoxide
N2O4 = Dinitrogen Tetroxide
Si2I6 = Disilicon Hexaiodide
Writing Chemical Formula
Compound must have charges that 'equal' out
Hydrates
Some Ionic compounds that attract water from water vapour in their solid state
Hydrated Compounds = w/ Water
Anhydrous Compounds = w/o Water
Examples
LiOH*H2O = Lithium oxide monohydrate
Na2CO3*10H2O = Sodium carbonate decahydrate
Cu(NO3)2*6H2O = Copper nitrate hexahydrate
Name compound, and then add the number of waters
Common Acids
HCl = hydrochloric acid
HBr = hydrobromic acid
HF = hydrofluoric acid
HI = hydroiodic acid
HNO3 = Nitric acid
H2SO4 = Sulphuric acid
H2SO3 = Sulphurous Acid
HNO2 = Nitrous acid
Hydrogen atoms = acid
Polyatomic Ions
Two or More
Types of Reactions
Synthesis
2 very simple things (usually elements) join 2gether to form 1 thing
A + B -> AB
Adding water to anhydrous compounds
Examples
2Cu(s) + O2(g) -> 2CuO(s)
2Sb + 3Br2 -> 2SbBr3
Decomposition
1 thing breaks apart to form 2 simple things (usually elements)
AB -> A + B
Removing water from hydrated compounds
Examples
2Ag2O -> 4Ag + O2
Replacement
Single
One thing is swapped with another thing
A + BX -> B + AX
Fe + CuSO4 -> Cu...