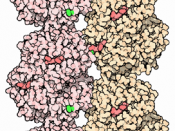
The objective of this experiment was to examine the electrophoretic behavior of hemoglobin from a normal person and from a person who has the sickle cell trait. Sickle cell disease is an inherited autosomal recessive gene. If the person has the inherited gene for sickle cell disease (hemoglobin S) from only one parent, then they are heterozygous and carry the trait. When the gene for hemoglobin S is inherited from both parents, the individual is homozygous and has a severe case of anemia. Electrophoresis is used in clinical laboratories to detect abnormal hemoglobins. Normal hemoglobin A had a strong negative charge therefore moved faster. Variant hemoglobin S had a weak negative charge resulting in slow movement.
INTRODUCTION
Red blood cells (erythrocytes) carry the globular protein hemoglobin in the circulation. This protein transports oxygen from the lungs to the tissue. The structure of hemoglobin has changed by mutations among human population.
One out of 100 people have a mutant hemoglobin gene and they have abnormal hemoglobin in their blood. Usually harmless, mutations involve the substitution of one amino acid for another; however, some mutations may cause serious diseases such as sickle cell disease. Hemoglobin S is the most common and serious abnormal hemoglobin. In this mutation, a single glutamic acid residue on the Beta-chains is replaced by valine. This single change in the primary sequence causes a marked change in net charge and conformation of the protein. When hemoglobin S is deoxygenated, it crystallizes in the red blood cells and this leads to a distortion of the red cells into a sickle shape. These abnormal cells are then destroyed rapidly in the body which leads to a reduced number of erythrocytes; hence the term, sickle cell anemia.
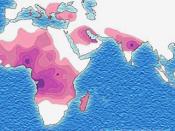
Hemoglobin S is thought to have originated in Africa among the Black population. It...