Objective:
The benefit of this lab was to acquaint oneself with the fundamentals of the Aldol Condensation reaction by demonstrating the synthesis of dibenzalacetone (trans, trans-1,5-Diphenyl-1,4-pentadien-3-one) through the aldol condensation of acetone with benzaldehyde. The synthesis began by using a strong base to generate the acetone enolate ion. The ketone/enol tautomerization is an equilibrium process that produces little of the enol (ppm or less). However, any enol that formed quickly reacted with the strong base to form the enolate ion. Therefore, the generation of the enolate drove this reaction to completion (Le Chatelier's principle). Being a very strong nucleophile, this enolate attacked the carbonyl of benzaldehyde and formed an alkoxide ion. This alkoxide ion abstracted a proton from water to form a beta keto alcohol which then tautermerizes to an enol. This enol reacted with base to form yet another enolate. Finally, the enolate lost a hydroxide ion to form an alpha, beta unsaturated ketone.
Since the newly formed ketone still possessed alpha hydrogens, it too underwent the same enolate condensation reaction with a second mole of benzaldehyde to form the final product, dibenzalacetone.
Background information was obtained on all aspects of this mechanism, fiddling with such concepts as equilibrium shifting, possible limitations, and symmetrical versus mixed aldols. The final product, dibenzalacetone, was weighed in order to calculate the percent yield. A melting point was then taken to properly characterize the final product.
Abstract:
The experimental procedure followed the format referenced in the lab manual Chemical Education Resources: Chem 236, Synt 720. The only experiment performed with the assistance of this lab manual was on page 101-103, Semi-Microscale Aldol Condensation.
Discussion:
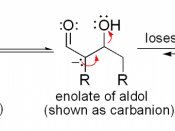
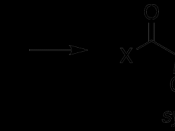
A majority of biomolecules, such as, carbohydrates, lipids, proteins, nucleic acids, and numerous others, are biosynthesized through pathways that involve carbonyl condensation reactions. Condensations are reactions that add together two or more molecules, often with the loss of a small molecule such as water or an alcohol. Carbonyl Condensation reactions occur this way. They take place between two carbonyl partners and involve a combination of nucleophilic addition and a-substitution steps. One component, the nucleophilic donor, is converted to its enolate and undergoes a a-substitution reaction. The other component, the electrophilic acceptor, undergoes nucleophilic addition. This general mechanism of carbonyl condensation is displayed below.
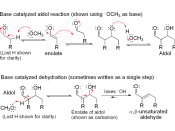
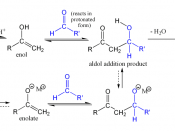
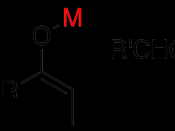
All carbonyl compounds, including aldehydes, ketones, esters, amides, and nitriles can endure condensation reactions. In this particular experiment, the carbonyl compounds were two aldehydes combined with an alcohol, otherwise known as an aldol. In its simplest form, acetaldehyde is treated with a base causing a rapid and reversible condensation reaction to occur. The product is a b-hydroxy aldehyde or ketone. This mechanism is known as an Aldol Condensation Reaction, a base-catalyzed dimerization of two aldehydes with a a hydrogen atom. This reaction only takes place if a a hydrogen atom is present. These hydrogens adjacent to the carbonyl are weakly acidic. Loss of these protons leads to a resonance stabilized enolate ion. The enolate ion is the strong nucleophile that attaches to the other carbonyl group giving a tetrahedral alkoxide ion intermediate. Protonation of the alkoxide ion intermediate yields a neutral aldol product and regenerates the base catalyst.
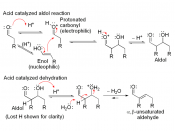
Aldol products can dehydrate under either acidic or basic conditions to give either a, b-unsaturated aldehydes. Even though Aldol condensation reactions are often evenly balanced equilibriums, the dehydration is usually exothermic (meaning heat releasing), driving the condensation to completion. The exact position of the aldol equilibrium depends both on reaction conditions and on substrate structure. Under basic conditions, an acidic a hydrogen is removed, yielding an enolate ion that expels the -OH leaving group. Under acidic conditions, an enol is formed, the -OH group is protonated, and water is expelled. Equilibrium can be shifted by the amount and strength of base added. If a full equivalent of strong base is added and the carbonyl compound is rapidly and completely converted into its enolate ion at a low temperature, with the addition of an electrophile to quench the reactive enolate ion, no condensation can take place. On the other hand, if we add a catalytic amount of weak base, rather than a full equivalent, a small amount of enolate ion will be generated. Once the condensation reaction has taken place, the basic catalyst is regenerated. For monosubstituted aldehydes, the equilibrium favors products, but for other aldehydes and ketones, the equilibrium favors the reactants. Steric factors are responsible for these developments, since increased substitution near the reaction site increases steric congestion in the aldol product.
As previously mentioned, aldol reactions are symmetrical, meaning; the two-carbonyl components are the same. If two similar aldehyde or ketones react under aldol conditions, four products may be formed. On the hand, aldol reactions can also be mixed, meaning an aldol reaction with two different carbonyl partners. A mixed aldol reaction leads to a mixture of products unless one of the partners either has no a hydrogens but is a good electrophile acceptor or is an unusually good nucleophilic donor. Therefore, the products used in mixed aldol reactions must be carefully selected to avoid unwanted product mixtures.
Summary:
Substance Quantity Molar Mass (g/mol) Melting Point (oC)
acetone 0.6 g 58.08
benzaldehyde 2.1 g 106.12
dibenzalacetone 224 110
ethanol 55 ml 46.07
Sodium hydroxide 10% 20 ml 40.00
Table 1 (SYNT 720 pg. 101)
Data:
Part 1
Weight of product: dibenzalacetone 6 mg
Part 2
Melting Points: Crude and recrystallized 111-112 oC
Part 3
Theoretical Yield
Part 4
Percent Yield
Conclusion:
In this experiment, acetone is used as the enolate forming compound, adding to the benzaldehyde followed by the dehydration to form a benzal group. The acetone has a hydrogens on both sides of the carbonyl group; therefore acetone can add two molecules of benzaldehyde. The condensation of acetone with the two molecules of benzaldehyde gives dibenzalacetone, otherwise known as 1,5-Diphenyl-1,4-pentadien-3-one.
The end product was recrystallized using ethanol. The final weight of my product was .006g. My percent yield was 10.3%, which some may say was less than desired. A melting point was then taken of both my crude product, as well as, my recrystallized product, resulting in identical melting points of 111-112oC, which indicate similar characteristics in my crude and