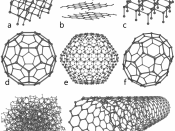
Allotropes are the existence of an element in more than one form. Carbon is one of the elements that contain many allotropes. Each allotrope of carbon has its own different properties. The allotropes of carbon are diamond, graphite, fullerenes, carbon nanotubes, carbon nanobuds, amorphous carbon, glassy carbon, lonsdaleite, and carbon nanofoam.
Diamond is the best know allotrope of carbon. It is the hardest known natural mineral which makes it very rough. In the structure of the diamond, each carbon atom is covalently bonded to four other carbon atoms and has a tetrahedral shape, meaning it is a nonpolar molecule. Diamond has a very melting point and has a density of 3.51 grams per cubic centimeter.
Graphite is the second allotrope of carbon. Its atoms of carbon are arranged in layers. Within each layer of carbon atoms, the atoms join to form six-membered rings and each carbon atom is connected to three others.
Two of the three bonds is a single covalent bond and the other bond is a double covalent bond. Graphite is very soft and slippery because the layers are not chemically bonded and can slide over the other. Graphite also has electrical conductivity and is less dense than Carbon since its density is 2.22 grams per cubic centimeter.
A third allotrope of carbon is the fullerenes. Each atom is bonded to three other atoms of carbon just like in the graphite structure, however, the atoms join to form both five-membered rings and six-membered rings. The chemical properties of fullerenes are still being researched by scientists and they are also studied for potential medical use in the belief that they can be put into specific antibiotics to target bacteria and cancer cells.
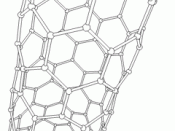
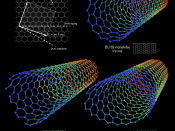
Another allotrope of carbon is carbon nanotubes. They have a cylindrical or circular cylinder shape. The name...