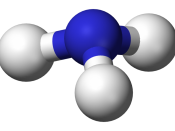
Ammonia (NH3) PROPERTIES This is a colourless, overpowering gas. A saturated aqueous solution of ammonia contains 45 percent ammonia by weight at 0ð C and 30 percent at ordinary room temperatures. On solution in water, ammonia becomes ammonium hydroxide, NH4OH, which is strongly basic and similar in chemical behavior to the hydroxides of the alkali metals. Ammonia melts at - 77.7ðC, boils at -33.35ðC, and has a density of 0.68 at its boiling point and 1 atm of pressure.
Ammonia is highly soluble in water and forms a solution known as ammonium hydroxide. Ammonia is not very reactive when dry, but it reacts with many chemicals when dissolved in water. Ammonium hydroxide neutralizes acids and forms the corresponding ammonium salts. For example, hydrochloric acid added to ammonium hydroxide produces a solution of ammonium chloride. When combined with some metals, ammonium hydroxide forms complex compounds called ammines. For example, the addition of ammonium hydroxide to a pale blue solution of cupric sulfate produces a deep blue solution of cupric ammine sulfate.
HISTORY Ammonia has been around for along time now dating back to the ancients. They had derived the name and the substance from sal ammoniac (a mineral that is one of the most common and most well known of the ammonium-bearing minerals). The ammonia was produced at the Temple of Jupiter Ammon in Libya by the distillation of camel dung. During the Middle Ages in Europe ammonia was obtained by heating the horns and hoofs of oxen and was called spirits of hartshorn.
USES Ammonia is widely used as a fertilizer. Ammonium nitrate and other ammonium salts help to increase crop production because they have a high percentage of nitrogen. In some farming areas, anhydrous ammonia is now applied directly to the fields from large tanks that contain the compressed gas.
Large quantities of ammonia are oxidized to make nitric acid, which is needed to make such explosives as TNT, nitroglycerin, and ammonium nitrate. The textile industry uses ammonia in the production of synthetic fibers such as nylon and cup ammonium rayon. Ammonia is also used in dyeing and scouring cotton, wool, and other fibers. Ammonia water sometimes serves as a cleaning fluid and can be used to restore fabrics that have been stained by acids.
Ammonia is also vital in the manufacture of many chemicals, plastics, vitamins, and drugs. For example, it acts as a catalyst in making plastics such as Bakelite, and it is used as a reactant in making melamine resins for plastics.
ANTIDOTE The common antidote to ammonia poisoning are drugs or other remedies that would counteract the effect of poisons. It may neutralize the poison chemically, as bicarbonate taken as an antidote for acid; it may render the poison insoluble and therefore harmless, as Epsom salt as an antidote for lead; it may absorb the poison, as charcoal as an antidote for alkaloids; or it may cost the stomach, as olive oil as an antidote for ammonia poisoning.