Lab:
Inhibiting the Action of Catechol Oxidase
By: Kimberly G.
Introduction:
In this lab, Mr. Greene's sixth period AP Biology class split into groups "to investigate inhibition of enzyme activity by specific chemicals called inhibitors" (1). Group three pondered this lab's inhibitor, phenylthiourea (PTU).
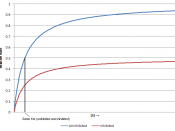
Is it a competitive inhibitor? That is an inhibitor that literally "competes" with the substrate by mimicking it, and thus "wins" the position at the active site of the enzyme. The blocked substrate in turn cannot join with the active site, causing the enzyme's activity to be hindered. This inhibition can be reversed, but only if the substrate's concentration levels are increasing raised while the inhibitor's concentration levels are remained stable.
Or is PTU a noncompetitive inhibitor? A noncompetitive inhibitor also joins with the enzyme, but it joins to a site other than the active site. This action changes the enzyme's nature causing the enzyme to lose its catalytic properties.
During this inhibition, the substrate cannot reverse the inhibitor's actions, so increasing the concentration of the substrate is fruitless.
PTU, an inhibitor of catechol oxidase, must join with copper, as a cofactor, in order for the catechol oxiadase to be active, and PTU is known for doing just that. Group Three hypothesized that PTU is in fact a noncompetitive inhibitor based on their knowledge of the nature of competitive and noncompetitive inhibitors.
The Group then went on to predict that there won't be a reaction when the substrate's concentration is raised because the Group predicted that it is a noncompetitive inhibitor.
Materials and Methods:
? Test tube rack
? 3 small test tubes
? Calibrated 5-ml pipette
? Distilled water
? Potato extract
? Catechol
? Disposable Pasteur pipettes
? Pipette bulb
? Phenylthiourea (PTU)
? Optional disposable gloves
Using Table 3.1 prepare...

This counts as an essay?
uh... I mean, cool lab man, but this paper is not across the border to essayville. And since we're not in your class, we don't really know what you're talking about w/o any background info. Sorry dude, but to me, this ain't an essay.
10 out of 11 people found this comment useful.