Atrazine
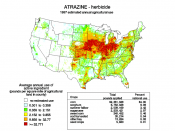
Atrazine is an herbicide used to control producers such as broadleaf and grassy weeds. Atrazine is mainly applied to corn and soybean crops, but is also used on sorghum, sugarcane, pineapple, and Christmas tree farms. This pesticide is a white crystalline solid organic compound that is available in many forms, such as a liquid, water dispersible granular liquid, and a powder. Trade names for atrazine include aatrex and atratol, and atrazine is in the family of triazines. The compound is both effective and inexpensive, and thus is well-suited to production systems with very narrow profit margins, as is often the case with maize. Atrazine treatments showed an average of 5.7 bushels more per acre than alternative herbicide treatments.
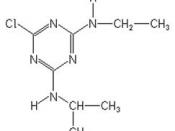
it melts at 448K and boils at 473K and is soluble up to 7mg per 100mL water. Atrazine is synthesized from cyanuric chloride which is then treated with ethylamine and isopropyl amine. Atrazine functions like the rest of the triazine family by bonding to the plastoquinone-binding protein which is not found in animals. Atrazine inhibits the electron transport process which causes starvation and oxidative damage and climaxes in the plant's death.
Atrazine was banned in the European Union (EU) in 2004 because of its persistent groundwater contamination. In the United States, however, atrazine is one of the most widely used herbicides, with 76 million pounds of it applied each year, in spite of the restriction that used to be imposed. Its endocrine disruptor effects, possible carcinogenic effect, and epidemiological connection to low sperm levels in men has led several researchers to call for banning it in the...