Avogadro's Number
Introduction:
Avogadro's number, is the number of "elementary entities" (usually atoms or molecules) in one mole. The mole is the SI unit for the amount of a substance and it is one of the seven fundamental SI units. It is defined as the amount of substance of a system which contains as many "elemental entities" as there are atoms. It is abbreviated "mol," and the number of entities in a mole of substance is given by Avogadro's number. The accepted numerical value of Avogadro's number is 6.02214199 x 1023, although generally when in use it is rounded to two or three decimal places. Avogadro's number is used extensively to calculate various volumes, masses, and numbers of particles involved in chemical changes and it is independent of the size or mass of the gas molecule. Avogadro's number, or the mole, is used in the same way that a dozen (which is twelve), or a score (which is twenty), is used.
Historical Background:
Avogadro's number, also called the Avogadro constant, is named after the early nineteenth century Italian scientist Lorenzo Romano Amedeo Carlo Avogadro, commonly known as Amedeo Avogadro, who in 1811, published a hypothesis stating that the volume of a gas is directly proportional to the number of atoms or molecules it has, regardless of the gas's nature. Avogadro based his work on the earlier discovery by Joseph Gay-Lussac that gases combine with each other in simple, whole-number ratios of volumes. Avogadro argued that the only way Gay-Lussac's discovery could be explained was to assume that one litre of any gas contains the same number of particles as one litre of any other gas. To explain this he further hypothesized that the particles of at least some gases consist of two particles bound together, a structure to which...

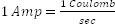
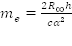
Avagadro's Number
This remarkably good and informative essay is marred by a weakness in the Cheathouse system, that it cannot show superscript or subscript annotation. Avagadro's number, "6.022 x 1023," is not "six point zero two two times one thousand twenty three." It is "six point zero two two times ten raised to the twenty-third power." Ten raised to the twenty third power is 100,000,000,000,000,000,000,000, a lot more than 1023.
1 out of 1 people found this comment useful.