Water is essential to life itself, with out water life on earth would not exist. Water is a major component of cells, typically forming between 70 and 95% of the mass of the cell. This means that we are made from approximately 80% water by mass and some soft bodied creatures such as jellyfish are made of up to 96% water. Water also provides an environment for organisms to live in, 75% of the earth is covered in water.
Water itself is a simple molecule made up of 2 hydrogen atoms and one oxygen atom, H20. The hydrogen and oxygen atoms are bonded covalently as shown in the diagram of water's electron structure. Water is not a linear molecule, the two hydrogen atoms form a bond with the oxygen at the angle of 104.5o.
Diagram of water's electron structure
Covalent bonds are formed by sharing electrons in the outer orbits of the quantum shells.
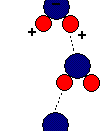
In the case of water however the large number of protons in the oxygen nucleus have a stronger attraction for these shared electrons than the comparatively tiny hydrogen nuclei. This pulls the electrons slightly closer to the oxygen nucleus and away from the hydrogen so that the oxygen develops a slight negative charge and the hydrogen's a slight positive charge. This makes the molecules slightly polar.
Diagram of water molecule showing slight charges
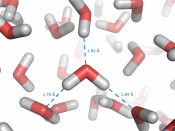
This slight charge means that when water molecules are close together the positively charged hydrogen atoms are attracted to the negatively charged oxygen atoms of another water molecule to form a weak hydrogen bond.
The bonds are weak individually but the sheer number of them means that the total force keeping the molecules together is considerable.
Diagram of water molecules forming hydrogen bonds
Water is an unusual substance, mostly due to it's hydrogen...