Organic Chemistry Classification
study of simple/synthetic/complex C-compounds
petroleum products and by-products: organic animal/vegetable, inorganic mineral (produced chemicals but now they are synthesized without having to extract, also carbonate & cyanide compounds)
6 common organic compounds in order of abundance from least to most: S, P, N, O, C, H
Bergelius' Vital Theory: forces produce compounds in living things only
Vohler, German scientist: 1824, produced oxalic acid COOH COOH & 1828 produced urea
other scientists began to synthetically produce acetic acid, benzene lab
Covalent bonds
Catenation: C atoms joined together in chain or ring/cyclo
London forces b/w organic molecules
Isomers: one molecular formula could represent more than one organic compound
Chemical properties determined by the functional groups
____________________________________________
Hydrocarbons
Non-polar, hydrophobic most
C bonds covalently with H
Branched, unbranched, rings, shapes, 1x/2x/3x bonds
Aliphatic: open chain, alkane C-C, alkene C=C, alkyne, cyclo, meth/eth/prop/but/pent/hex/hept/non/dec
Aromatic: benzene ring, have aroma (scent) ie. Benzene C6H6, see Kekule Hydrocarbons below
C-H bonds consistent length with C-H bonds of hydrocarbons (C-C > C-_-_-C bond length < C=C)
All C-C have same length/reactivity = structural formula written as I/II/III "Resonance Formula"
____________________________________________
Polar
soluble in water, hydrophilic (alkanes/parafines)
CO: diatomic (2 atoms of same/different elements)
single H (HCl), OH (C2H5OH), O (OCl), N (NF3)
alcohol (hydroxyl), aldehyde/ketone (carbonyl), carboxylic acid (carboxyl), amine (amino)
Non-Polar
insoluble in water, hydrophobic (alkenes/ynes)
all elements Cl2
most C compounds: oil, CO2, CH4
____________________________________________
Saturated Hydrocarbons
single C-C bonds, alkanes/parafines
Max # of H atoms
Fats
Substitution reaction
Unsaturated Hydrocarbons
Double C=C or triples bonds, alkenes, alkynes, non-polar
Oils, Teflon, Plexiglass (contacts), Synthetic rubber (Cl radical), Natural Rubber (methyl radical), tire rubber, (black since C-S not C-H monomers, smell since Sulphur burns)
Addition reaction
Polyunsaturated
1+ 2x bond
Polymers
Chain molecules of identical monomer units (held by Van der Waals Forces)
Polysaccharides, plastics
Monomers
smallest repeating unit of polymer
monosaccharides
1 molecule = dimer, 2 molecules = trimer, 3 molecules = polymer
1000s of monosaccharides linked in branched or unbranched (linear) chains
Hydrophilic: water loving
Hydrophobic: water hating (fats, hydrocarbons, oils, waxes)
Amphiliphic: dual nature (phospholipids)
____________________________________________
PROPERTIES OF H2O
High surface tension/cohesion
leaves pull upward from root
High boiling point
wants to stick together from hydrogen bonds
lot of energy to break H-bonds
liquid into gas state (hard to boil into gas)
Temperature stabilizer
lots of energy
H-bonds absorb heat
H-bonds break and release heat
raise temperature of water (otherwise moderates temperature changes near large bodies of water)
Ice less dense then water
water cools
molecules have less kinetic energy
H-bonds get stronger & hold water molecules in rigid crystalline structure
ice volume more than liquid = ice lower density = ice floats to top...life
gone if lakes froze into ice water
____________________________________________
Carbon Compounds
Hydrocarbons largest group, most have carbon-carbon covalent bonds, aliphatic & aromatic
Long chains of atoms that are covalently bonded to hydrogen atoms and other carbon atoms
Non-polar, almost all hydrophobic (water-hating)
Branched/unbranched(linear), chains/cyclic(ring) shapes, 2x/3x bonds, isomers
Bohr-Rutherford Diagram of Carbon: 6p, 2e, 4ve
____________________________________________
ISOMERS
same #/type of atom but different arrangement
Structural
different covalent arrangement b/w atoms
different loc. of double/triple bonds
Geometric
same covalent arrangement b/w atoms
inflexibility of 2x bond, no rotation possible as with C-C
Optical/Enantiomers
mirror image, D - amino acids & L - amino acids
inactive/harmful (thalidomide sedative toxic to embryo & fetus)
____________________________________________
Structural Formula: H3-C-C-H3 or H3CCH3
Molecular Formula: C2H6
Radical: CH3 (methyl) radical of CH4 (methane) & C5H11 (pentyl) radical of C5H12 (pentane)
Atoma: indivisible
LINKAGES
Ether
b/w alcohols, R - O - R
polar, but not as polar as alcohols
boiling point lower than alcohols, good solvent
Ester (scent)
b/w alcohols and carboxylic acids R - (C=O) - O - R
found in hydrocarbon chains in phospholipids, mono/triglycerides
esterification, hydrolosis
Organic Acids: R - C=O - OH
Cyanides: R - C -3- N, R - C(=-)N
Sulphonates: R - SH - O3
Anhydrides: b/w acids
Peptides: b/w amine & acids
Pi Bond
Double bond
2 electrons shared by overlapping orbital, tend to arrange so mix overlapping of the orbitals occur
Hybrid orbital: electron from one orbital moves into an empty orbital but on the same energy level =equals= an extended valence =then= can bond with more atoms
Double Bonds: C=C-C-C-C
Conjugated: C=C-C=C-C=C, alternating
Cumulate: C=C=C=C=C=C
Sigma Bond
single
overlapping of s/p/sp orbitals =makes= bond (where electron density concentrated above/below imaginary rest line)
electron density b/w nuclei of 2 atoms along imaginary rest line
____________________________________________
CARBONYL
polar
role in carbohydrate formation: precursor for simple sugars, oxidizes readily to form carboxylic acids
oxidation, hydrogenation
Aldehyde
general formula: R - CH=O (no isomers)
simplest: CH2O, formaldehyde
"-al" IUPAC ending (International Union of Pure and Applied Chemistry)
Ketone
general formula: R - (CH=O) - R
simplest: CH3COCH3, acetone/dimethyl ketone
"-one" IUPAC ending (International Union of Pure and Applied Chemistry)
found in fats
HYDROXL
Alcohol
polar
general formula:
Primary: R - OH, d.brown
Secondary: R - OH - R, brown
Tertiary: R - OHR - R, purple
simplest: CH3OH, methanol
"-ol" IUPAC ending (International Union of Pure and Applied Chemistry)
Phenol: product when -OH is added to an aromatic ring
CARBOXYL
Carboxylic Acid
polar
general formula: R - C(OH) =O
simplest: CHOOH, methanoic acid
"-ic" IUPAC ending (International Union of Pure and Applied Chemistry)
in amino acids and fats, highly reactive, produce H+ ions in soln
2 dicarboxylic acids: 1--- COOH COOH, oxalic acid 2---COOH (CH2)3COOH, succinic acid
ionization, neutralization, reacts with metals, esterification
AMINO
Amines
polar, weaker since H has low electro-negativity
general formula: R - C - NH2
simplest: CH3NH2, methyl amine
"-amine" IUPAC ending (International Union of Pure and Applied Chemistry)
in amino acids: enables it to act as base, removes H+ from solution, form OH- ions
Amides
general formula: R - (C=O) - NH2
CH3CONH2, ethanamide
"-amide" IUPAC ending (International Union of Pure and Applied Chemistry)
SULPHIDE
Thiol
slightly polar
general formula: R--SH
simplest: CH3SH, methathiol
"-thiol" IUPAC ending (International Union of Pure and Applied Chemistry)
PHOSPHATE
Phosphate *not really organic*
Polar
General formula: R-O-P-(=O, OH, OH)
Part of DNA & RNA so it is abundant since everything has DNA
____________________________________________
Electronegativity (Linus Pauling)
to attract shared electron pairs when forming bonds (like to be stable so pair up & fill outer (valence) shell)
Depends on # of protons and size of atom (distance from valence to the nucleus)
Increases as we go up/across the periodic table (ie. F is most electro (-) element)
Determines type of bond that forms b/w two atoms
____________________________________________
Van der Waals forces (strongest >>> weakest)
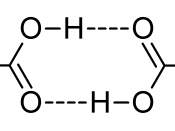
Hydrogen Bonds: b/w polar (H & F O, N) ~~~ covalent bonds: H very + and others - (H2O, NH3, HF, CH3OH)
Dipole-dipole: b/w polar ~~~ + attracted to - charged (SCl2, PCl3, CH3Cl)
London/Dispersion forces: b/w non-polar molecules (H2, Cl2, CO2, N2O4, CH4) >>>uneven e- distribution in close molecules, more e- = stronger dispersion force
INTRAMOLECULAR FORCES (Van der Waals forces 1 & 2)
Electron cloud size: large = not held tightly = forces stronger
# of atoms: more atoms = more forces = higher temperature
Ionic Bonds
Metals (LS of PT) & Non-metals (RS of PT)
Strongest bond: form b/w atoms w/ different negativities
Na + Cl Na+[Cl]-, unequal sharing of electron pairs: more electromagnetic atom will attract/steal electron pair becoming -ly charged ion, while the other becomes +ly charged
Have high melting points, dissociate (split apart) in aqueous solution
Covalent Bonds
common in organic life forms, bonds b/w atoms of similar/identical electro negativities, high electron similarity/ionization energy
high melting/boiling points, remain liquids over short temp range, don't conduct electricity
Bond Length: Distance of covalent bonds, C--_--_--C length b/w C-C and C=C (gives substitution not addition)
Bond Energy/Enthalpy = bonds broken - bonds formed
2 atoms approach
e- density around each nucleus shifts to the middle
distance decreases, probability of finding either e- near their nucleus until each of H-atoms in molecule reaches a sharing of the two e-
atoms of different elements form compounds (classical naming system: iron + sulfur = iron sulfur)
3 Types of Covalent Bonds (strongest >>> weakest):
Polar Covalent: different electro negativities, H2O
Non-Polar/Pure Covalent Bonds: similar/identical electro negativities, O2
Coordinate Covalent: shared electrons donated by same atom, weak since borrow lone pair, SO2 & H3PO4
Why Some Covalent Compounds Dissolve In H2O?
C + H atoms = polar, small difference in e-negativity level
O + H atoms = polar, ie. H2O + alcohol
(+) dipole + (-) dipole = Hydrogen bond, attracted to each other
INTERMOLECULAR FORCES (Van der Waals forces 1, 2 & 3)
Weaker than covalent bonds: b/w electrostatically attracted molecules
RNA & DNA
2 important pentose sugars, many phosphate groups
DNA - Deoxyribose: C5H10O4 (missing -OH group)
RNA - Ribose: C5H10O5
Lipids
in phospholipid cell membrane
consist of hydrocarbons in ester linkage
C, H, more carbohydrates then O's
4 uses: Protect organs (kidneys), Prevent H2O loss in plants, Long term energy, Thermal insulation (bladder)
Steroids/Sterols
17 C atoms in 4 rings
testosterone/estrogen (sexual), adrenaline (heart), anabolic (muscular), bile salts (digestive)
Waxes
Fatty acids + monohydroxy alcohol
Hydrophobic, high melting points
Protective outer covering of fruits, leaves, animals skins
Glycerides
Glycerol: 3C with OH on each (trihydroxy alcohol) + fatty acid's COOH (carboxyl) in synthesis/condensation rxn = ester link
Monoglyceride: 1 fatty acid + glycerol in dehydration rxn = ester linkage
Diglyceride: 2 fatty acid + glycerol in dehydration reactions
Triglyceride: 3 fatty acid + glycerol in dehydration reactions
Oils: unsaturated, liquid, hydrophobic, low melting point, olive oil
Fats: saturated, solid @ room temp, hydrophobic, high melting points, butter fats
Fatty acids: long hydrocarbon chains (unbranched) ending in COOH (monocarboxylic), even #of C (16-24 C)
Glucose
C6H12O6 (1:2:1) aldohexose (6C)
16 isomers (mainly stereoisomers) + 5 -OH groups + C=O at end
1 isomer of glucose: fructose, C6H12O6 (1:2:1)
1 stereoisomer of glucose: galactose, C6H12O6 (1:2:1)
____________________________________________
Chlorophyll A
Participates in light rxns
Head: porphyrin ring w/ Mg center
Tail: hydrocarbons, interacts with hydrophobic proteins in the thylakoid membrane
Carbonyl group: CH3 bonded to porphyrin ring (head)
Chlorophyll B
Methyl group: not CH3 but CHO bonded to porphyrin ring (head)
Carotinoid
Yellow to red pigments (as carotenes)
2 small 6C rings, connected by chain of C atoms
REACTIONS
Acid-Base Reaction
Involve transfer of protons [H]+ from one molecule to another
Acids donate [H]+, bases accept
Oxidation-Reduction 'Redox' Reactions
Involve transfer of electrons from one molecule to another
Reducing agent stays the same in reactants and products, oxidizing agent is split apart in product
Condensation (dehydration, condensation, synthesis):
Hydrolysis Reaction (reverse of synthesis: hydrolysis, degragation, digestion):
H2O produced/used to break/form new bonds
These bring monomers together to form bond
Phosphorylation
Carboxylic acid + water carboxylic acid + alcohol
ATP + H2O ADP + Phosphate
Transfer Pi b/w mol, if added endothermic & lost exothermic
Decarboxilylation
COOH removed, releases O2
Catalyst can be NaOH
Isomerization
Substrate into isomer
Oxidative Phosphorylation
Product is pyruvic acid
Pyruvate Oxidation
In mitochondrial matrix
2 pyruvic molecules produced at the end of glycolysis
Get converted into Acetyl CoA by addition of coenzyme A
Oxidative Decarboxylation
Decarboxylation + redox reaction
Oxidation
Lose e- and H atoms
Combustion: Oxygen, water is product
Reduction/Hydrogenation
Gain e-/ H atom
NAD, FAD coenzymes accept e- from substrate
C=C-C C-C-C
Hydration
Add water to break a bond, such as double bond in alkenes
CH2=CH2 -----> H2O, [+H] ----> CH3-CH2-OH
Ethane -----> H2O, [+H] ----> ethanol
Fermentation
CO2 is released
Thermic Decomposition
Cracking: alkanes to alkenes, TÃÂC @ 650ÃÂC
Pyrolise: compound is split, TÃÂC > 1000ÃÂC
Elimination
Alkanes produces alkenes, water and [H]+
When between 2 monomers makes a condensation reaction
CH3CH2OH2+ CH2=CH2 + H2O + [H]+
Ionization
Acid is added to water, products are -charged acid radical and +charged H3O
Acetic/ethanoic acid acetate/ethanoate (acid radical), CCOOH + HOH CCOO- + HHOH+
Esterification
Ethanoic acid + methanol methyl ethanoate, or dimethyl ester + water
Substitution/Halogenation
Usually occurs with Fe and Br2
Chain Reaction: CH4 + Cl2 CH3Cl + HCl
Alkenes, industrial field
add halogens
Addition
Use Markovnikov Rule
Hx(aq) = hydro...ic acid
Wohler Method
(C=-C) both C attached to one Ca water vapour is added Ca(OH)2 + (C=-C)
___________________________________________
REACTIONS
A - Element hard to separate, 1 atom
B - Binary compound
C - Compound 2+elements chemically united
Mixtures: easy to separate, combination of substances
Metals: form + ions (cations), copper
Non-metals: form - ions (anions), O2
Electrolytes: conduct electricity
Oxides: O2 + element, H20
Synthesis:
Element + element = binary compound
A+ A=C
Hydrogen + Chlorine = Hydrogen Chloride
Decomposition:
Binary compound = element + element
C = A + A
CH4 + O = CO2 + H2O
Single-Displacement:
A1 + C1 = A2 + C2
Zn + H2SO4 = ZnSO4 + H2
Double Displacement:
C1x + C2y = C1y + C2x
NaCl + NgNO3 = NaNO3 + AgCl
Ag + Cl = AgCl
___________________________________________
Thermodynamics: study of Energy in matter
Efree = Etot + Entropy (Entropy is measure of chaos in universe)
âÂÂG= âÂÂH + Entropy x âÂÂT
Epot: stored
Ekinetic: E of motion
Eheat: random motion of molecule, wasteful, can't do work with it
Energy neither created/lost, but transferred/transformed
Energy transfer = increase in Entropy
Endothermic (ÃÂG='+')
heat made
ATP lost, Pi gained
entropy increases
Anabolic: small > large polymers w/ energy
Exothermic (ÃÂG='-')
heat lost
ATP gained, Pi lost
entropy decreases
Catabolic: large > small
___________________________________________
Metabolism
chemical reaction in organisms
Cellular Respiration
glucose 3 molecules of CO2
cytochrome proteins (in mitochondrial membrane) pass electrons to O2 (product of C. Respiration)
molecular oxygen is reduced
___________________________________________
Photosynthesis
Solar energy > saccharides synthesized > inorganic components > potential chemical energy
6CO2 + 6H2O > C6H12O6 + 6O2
Green bacteria: cellular membrane (no organelles)
Plants: chloroplasts
Occurs through two reactions
1. Light-Dependent Reactions:
Within grana
Light + H2O = ATP + NADPH
Require light directly
Solar energy >>> chemical potential energy
Recycle waste products of Calvin Cycle
2. Calvin Cycle/Light-independent reaction
Within in stroma
Require light indirectly
Chemical to ATP + NADPH+ CO2 = sugar (CH2O)
Chemical potential energy used to drive anabolic reactions that create energy-rich sugars
Depends on products of light-dependent reaction to occur
Non-Cyclic Electron Flow
Protons, photons (light) hits thylakoid membrane
Hits antennae complex
Hits light-trapping pigment (Ch. A, Ch.B, Cartenoids)
Trap light and transfer energy from molecule to molecule
Reaches reaction center and absorbs energy
Energy absorbed drives oxidation reaction
Reaction donates excited electron to the primary electron acceptor (redox reaction) through e- carriers
H2O excites & oxidizes (looses 2 electrons)
Result is 2H+ + O2
2H plus picked up by reaction center
Electron replaces ones lost in step 2 to primary receptor
2H plus in cytochrome complex produced (keep H+ high)
2H plus undergoes series of redox reactions (cytochrome proteins)
Cytochrome proteins use energy of electrons to pump H+ into thylakoid compartment (keeps low H in stroma)
Protons diffuse out through ATP synthetase
ATP synthesized
Excited electrons passes through series of enzymes
Used by NADP reductase (enzyme), NADP+ >>> NADPH (chemical potential energy)
NADPH (chemical potential energy) used in Calvin Cycle to take CO2 (non e-rich) and build it into sugar
DNA Structure
Found in nucleus of all dividing cells, carries genetic material
Open structure, 2x stranded helix that twists
Held by H bonds between nitrogenous bases (+ivly charged, hydrophobic)
-ivly charged phosphates like to stay outside and interact with +ivly charged/aqueous/polar/hydrophilic nucleus environment
H2O content high, but when low H2O content then bases tilt and compact
2 Helical Chains/Strands
Sequence of atoms in opposite directions
Follow right-handed helixes, coiled around same axis
Held together by 2 bases perpendicular to fibre axis
On each strand there is a 5' and 3' end
Phosphodiester Bonds
Joins polymer of nucleotides
Between the 3 OH (on the DNA sugar) & the phosphate group (on the adjacent nucleotide)
Bases
Joined by hydrogen bonds
Lie side-by-side = z coordinates identical
Specific bonding pairs so spread out evenly = no gaps/kinks/errors in DNA
2 types of bases
A (2x bond) T, purine position 1 + pyrimidine position 1
C (3x bond) G, purine position 6 + pyrimidine position 6
Purines
Adenine, guanine
Larger, 2x ring
Pyrimidine
Thymine, cytosine
Smaller, 1x ring
Acids (produce H+ ions)
taste: sour (acere = Latin "sour")
changes litmus: blue >>> red
*conducts electric current: H2O solns conduct (electrolytes)
react with: bases to form salts + H2O (neutralization reaction)
other: evolve H2 gas when rxn with active metal (Alkali mtls, zinc, aluminium)
examples: citric acid (fruits, veggies), ascorbic acid (vitamin c), vinegar (5% acetic acid), lactic acid (buttermilk), Sulphuric Acid
Bases (remove H+ ions)
taste: bitter
changes litmus: red >>> blue
*conducts electric current: H2O solns conduct (electrolytes)
react with: acids to form salts and H2O (neutralization reaction)
other: feel slippery/soapy
examples: detergents, soaps, lye (NaOH), household ammonia, Sodium hydroxide
Salts (produce non-H+/OH-ions)
taste: salty
examples: Sodium chloride
Homeostasis
maintain constant internal environment, steady state
recognize and respond to stress/significant change
External Stress: change in light, temp, food, produce
Internal Stress: metabolic activities, water, substrates disease, aging
Stress
stress produced (controlled by feedback mechanisms)
series of reactions
response/feedback: +/- (+ reduces stress, - increa stimulus)
Feedback System
receptors - detect stimulus
regulators - course of action/response
effectors - execute response
Sensory pathway: receptors > regulators
Motor pathway: regulators > effectors
Homeostatic System
1. Limits:
Boundaries, responds to critical amounts of stress, cannot cope with extreme envio. changes)
thermoregulation only effective in 15ÃÂC - 50ÃÂC, beyond range homeostatic system can't cope = hypothermia
2. Energy
energy to activate effectors, effectors activate large amounts of ATP
What is Homeostatically Maintained?
Nutrient Concentration (supply of energy production)
O2 [] (needs for energy production)
CO2 [] (in excess leads to acid formation in blood)
pH (changes outside normal range can affect enzyme activity and nerve cell communication)
Salt/electrolytes [] (maintain proper volume of cells, role in cardiac function/nerve cell communication)
Temperature (nerve cells detect temp 2 low=cell function slow, temp 2 high= structural/enzymatic proteins are impaired)
Volume & pressure (plasma volume-pressure ration to ensure body-wide distribution)
Reflexes
Mammalian Diving Reflex: slight changes in infants can damage enzymes and cause brain damage
Hypothermia: body mimicking sleep heart rate shoots up in cold water because of shock, but then comes down to conserve heat
Achilles Reflex, Knee Jerk: action of walking, used to that motion
Babinski Reflex: toes curl, motor functions, sensitive nerve
Papillary Reflex: pupils dilute and expands with increase/decrease of light
Aldosterone in Osmoregulation:
A steroid hormone (regulates Na+ and K+) secreted by adrenal gland
controls cells of distal tubule: promotes (secretion of K+ from blood>distal tubule) & (absorption of Na+ from distal tubule>blood)
high K+ in blood >>> increased aldosterone secretion >>> secretion of K+ into distal tubule from blood >>> reduced K+ in blood
___________________________________________
Diabetes Mellitus
'diabetes' to walk or stand with legs apart & 'mellitus' sweet pee
disorder in which you cannot use the nutrients you digest properly, especially sugar (stays in bloodstream,
goes into urine)
Insulin
takes sugar out of blood stream and makes energy, diabetics do not have the insulin to extract this energy
HYPOglycaemia: too little sugar, may pass out, not enough energy, may go into coma
HYPERglycemia: too much sugar in blood, thirsty, nap, need to pee, blindness (blood clots in eyes), kidneys shut down, amputation (circulation, cells bounce off in tight space and damage cells)Type I: 10% of cases
body cannot produce insulin
auto-immune disorder/juvenile diabetes: at birth or before birth or at young age body makes mistake, says beta cells are bad and foreign and kills them all off, therefore body cannot make insulin
Treatment: violet cell implants ie. cells that make diabetes, insulin injections
Type II: 90 % of cases, 1 in 4 chance
body does not make enough insulin or is inefficient at using insulin
genetic/environmental/adult-onset diabetes, inactivity and obesity, do not exercise , strain on pancreas, body cannot distribute or create insulin
Treatment: pills and exercise
Diabetes Insipidus:
damage hypothalamus, which makes NADH, which makes nerve cells
urine input increases so drink more water
Kidney Stones
more concentrated urine (minerals, alkaline and acid salts)
abdominal lower back pain, more urine output, tears tissues, causes pain so must be surgically removed
Bright's Disease
inflammation of nephrons and glomerulus lets through larger molecules that should not be there
urine output and urgency increases, thirst so drink water
Kidney/Bladder Cancer
smoking, exposure to cadmium, hereditary (as with all cancers)
pain, fatigue (growth of cells taking nutrients), blood in urine, urine output increases so treat with chemo, amino therapy, kidney transplants
Interstitial Cystitis
'itis' means inflammation
frequent urination, discomfort, blood in urine, so urinalysis, steroids, radiation, cystoscopy
Very Nice Work
These notes are very thorough and cover many topics in Biology, the only thing I would suggest is a little better organization but overall very good!
0 out of 0 people found this comment useful.