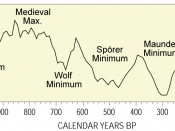
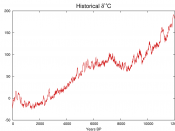
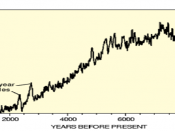
Abundance of Carbon 14 in the atmosphere=(0.0000000001%) or 1 part per trillion The percentage of carbon in the atmosphere that is Carbon 14 is a trace amount measured at .1% Carbon 14 is created in the lower stratosphere and upper troposphere and is then mixed with water to create carbon dioxide. This carbon dioxide is then absorbed by plants through photosynthesis and then animals eat the plants meaning that every living thing on the planet contains carbon 14. A radioisotope is a radioactive isotope of a certain element. Elements are substances that make up everything in our universe. Most elements are stable meaning that they do not want to break down and that they are happy to just stay in the from they are in. An Isotope is a different strand of the same element. It is a different strand because it has a different number of neutrons to the base element. If you have more neutrons than protons in a small enough space it will cause the isotope to become unstable therefore breaking down. When an unstable element breaks down it is known as radioactive decay. Therefore it is a radioisotope. A half life is the amount of time it takes for half of the atoms in the radioisotope to decay through nuclear decay into a stable element. For example if you had 50 atoms of Carbon 14 and it's half life is 10 seconds then on average (as there is natural variation in this process) after 10 seconds there will be 25 carbon 14 atoms left and so on. The Radioactivity of the radioisotope carbon 14 is the best thing for dating once living objects because every living object on this planet contains carbon 14. The way carbon 14 is used to date objects is that you have...
More Chemistry
essays:
Carbon 14
... half is known as the half-life of carbon-14. "The measurement of the remaining proportion of 14C in organic matter thus gives an estimate of its age, a raw radiocarbon age" (Staff of Beta Analytic Inc, N.D.). After decaying ...
Ragweed
... (French physicist) 499. Known as the discoverer of radioactivity. Reported in 1896 his observation that potassium ... much more realible than other standards of dating artifacts. Lindemann, F.A. -- (American chemist), 465 ... , Lothar -- (German chemist), 22-3 The second half of the pair of chemists to ...
Scientenstences
... (French physicist) 499. Known as the discoverer of radioactivity. Reported in 1896 his observation that potassium ... much more realible than other standards of dating artifacts. Lindemann, F.A. -- (American chemist), 465 ... , Lothar -- (German chemist), 22-3 The second half of the pair of chemists to ...
“an Overview Of The Current And Future Importance Of Oil In The World In Terms Of Energy And Resources”
... not, are neither technically possible nor economic to produce. Prospective Resources: quantities of oil and gas estimated on a given date to be potentially recoverable from undiscovered accumulations. Add to this the general attitude of the OPEC countries to give misleading ...
Chemical Processes - Iron Ore, Pig Iron, Wrought Iron and Steels
... /Carbon_steel 12 Chemistry Chemical Process Research Report Iron ore, Pig iron, Wrought iron and Steels Ben Cotterill Teacher - Mr Cochran Date due 3/8/07 - Increased strength, hardiness and brittleness - Reduced malleability, flexibility, ductility, and melting point - Reduced strength, hardiness ...