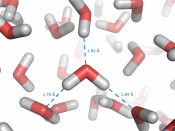
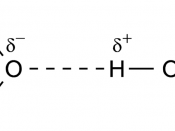Equilibrium-reversible process - a process that may occur in either direction (conversion of reactants to products and conversion of products to reactants, and it occurs simultaneously)1) Changes of StateH2O(l) H2O (g)1. When you first pour the water in the flask, there's not much water vapor above the liquid. Over time, molecules will evaporate and enter the space above the liquid.
2. All the liquid will NOT evaporate. There is not enough space in the air above the liquid to accommodate all of those water molecules. Eventually the space above the liquid will become saturated with water vapor.
3. If a vapor molecule collides with the surface of the liquid, it will re-condense. One is re-forming the IMFs that you once broke (heat of vaporization - takes energy to break the bonds in order to transform into the vapor state)4. Now equilibrium is established. The air is saturated with vapor and the rate of molecules leaving the liquid is the SAME as the rate of the molecules entering the liquid.
The amount of liquid and amount of vapor is going to stay constant.
has to reach a point of saturation before the vapor re-condenses (this is why it will not work if the cap is not on)-there is much more air now and it will still not become saturated once all the water has evaporatedSaturated solution - solute sitting at bottom-CaCO3 (s) Ca2+(aq) + CO22-(aq)-Calcium ions and carbonate ions will likely collide with the solid solute at the bottomand re-crystallize. At the same time, some of the solid solute may dissolve into the solution.
-energy being used to break the bonds is used to form them (no net energy change)-the RATES of dissolving and re-crystallizing are equal - solution equilibrium occurs in a saturated solution, both with solids and gasesSolution...