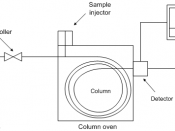
IntroductionThe purpose of this experiment is split up into three parts: separating cyclohexane and toluene through distillation, performing gas chromatography on the substances that were distilled, and separating a salt-water mixture through distillation. Distillation involves the ideas of vaporization and condensation. Through these procedures, liquid mixtures can be purified. An important step in the process is finding a boiling point. When liquids are heated, there is an energy added to the system. Once enough energy has been added, a molecule can change its conformation (liquid to a gas). Once a molecule changes from the liquid phase to the vapor phase, the vapor exerts a pressure called equilibrium vapor pressure. The more molecules that convert to gases, and how temperature is controlled, will cause this pressure to increase. Boiling begins when the equilibrium vapor pressure equals the pressure given off by the atmosphere, this is the boiling point.
In order to understand boiling points, the ideal gas law (PV = nRT) can be used.
Boiling points are measured after temperature and pressure have been figured out. Pressure in a system can adversely affect boiling point, therefore all calculations will be taken at Standard Temperature and Pressure (STP), 760 mm Hg. One warning to note is never heat a closed system because glassware could break. Instead of heating, a vacuum will be used to lower the pressure and in turn lower the boiling point. This procedure helps when samples are unstable at high temperatures.
There are four main types of distillation. One is simple distillation, the separation of liquids that boil below 150 ðC at 1 atm from non-volatile impurities and other soluble liquids that have a boiling point at least 25 ðC higher than the first. When performing a simple distillation, everything must be done correctly. All glassware needs...