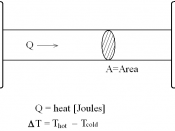
Does warm water freeze faster than cold water?
Experimental evidence seems to indicate that hot water does indeed freeze faster than cold water - this seems counterintuitive and there are a number of different explanations for the phenomenon. I don't think anyone has found a grant to study it in depth so there are not clear answers to the cause.
Thermal contact argument
The hot water melts the ice under the tray and when it refreezes there is more surface area in contact with the ice tray. Air is a reasonably bad conductor of heat - water or ice are much better so if you have a solid layer of ice touching the ice tray any heat is conducted away more quickly. Even when the hot water cools to the cold water temperature it will retain this layer of ice and so freeze solid more quickly. In order to see if this is true it would be necessary to put the tray in a completely frost free freezer or somehow hang it from the top!
Reduction in volume argument
People who have done experiments on this argue against the case that the volume of the hot water is reduced by evaporation, and therefore less hot water freezes more quickly than more cold water.
It is one argument but it isn't popular.
Convection current argument
From a New Scientist Last Word question, someone who did the experiment claimed that the longest time for freezing was at 5C while the shortest time for freezing was at 35C. This is an extract:
"The rate of heat loss from the upper surface is proportional to the temperature. If the surface can be kept at a higher temperature than the bulk of the liquid, then the rate of heat loss will be greater than from...