15.1 Electric Charge and the Electrical Structure of Matter
Fundamental Law of Electric Charge:
Opposite electric charges attract each other. Similar electric charges repel each other.
A Simple Model of the Structure of Matter
1. All matter is composed of sub-microscopic particles, called atoms.
2. Electric charges are carried by particles within each atom that are called electrons and protons.
3. Protons are found in a small central region of the atom, called the nucleus. They are small, heavy particles, and each one carries a positive electric charge of a specific magnitude, called the elementary.
4. Electrons move about in the vast space around this central nucleus. They are small, very light particles, yet each of them carries a negative electric charge equal in magnitude to that of the proton.
5. Atoms are normally electrically neutral, because the number of positive protons in the nucleus is equal to the number of negative electrons moving around the nucleus.
6. Neutrons are small, heavy particles found in the nucleus, and they carry no electric charge.
7. An atom may gain electrons. Then it is no longer neutral, but instead has an excess of electrons and, therefore, a net positive charge. Such an atom is called a negative ion.
8. An atom may also lose electrons. As a result, it will have a deficit of electrons and, therefore, a net positive charge. Such an atom is called a positive ion.
All electric charges in solids result from either an excess or a deficit of electrons.
Conductor- solids in which electrons are able to move easily from one atom to another.
Insulator- solids in which the electrons are not free to move about easily from atom to another.
Gazes and liquids can be either conductors or insulators.
15.2 Transfer of Electric Charge
Induced Charge Separation
The positive charges on a solid conductor are fixed and cannot move. Some negative electrons are quite free to move about from atom to atom.
Charging by Contact
An object charged by contact has the same sign as the charging rod.
Charging by Induction
An object that is charged by induction has a charge opposite to that of the carrying rod.
15.3 Electric Forces - Coulomb's Law
The magnitude of the electric force is inversely proportional to the square of the distance between the centers of the charged objects.
15.4 Electric Fields
Every charged object creates an electric field of force in the space around it. Any other charged object in that space will experience a force of attraction or repulsion from the presence of the electric field
15.5 Electric Potential
1 V is the electric potential at a point in an electric field. If 1 J of work is required to move 1 C of charge from infinity to that point.
15.6 The Milikan Experiment - Determination of the Elementary Charge
e = 1.602 x 10 C q = Ne
16.1 Natural Magnetism
Magnets
Law of Magnetic Poles:
Opposite magnetic poles attract. Similar magnetic poles repel.
Induced Magnetism: Temporary and Permanent Magnets
This model provides a simple explanation for many common properties of induced magnets:
1. A needle can be magnetized by stroking it in one direction with a strong permanent magnet, thereby aligning its domains.
2. When a bar magnet is broken in two rather than producing separate north and south poles, two smaller magnets are produced, each with its own north and south poles.
3. Some induced magnets made of soft iron demagnetize instantaneously, while others made of hard steel or alloys remain magnetized indefinitely. Impurities in the alloys seem to 'lock' the aligned domains in place and prevent them from relaxing to their random orientation.
4. Heating or dropping a magnet can cause it to lose its magnetization, jostling the domains sufficiently to allow them to move and resume their random orientation.
5. A strong magnetic field can reverse the magnetism in bar magnets so that the pole marked S points north. This occurs when the domains reverse their direction of orientation by 180 due to the influence of the strong external field in the opposite direction.
6. Ships' hulls, columns and beams in buildings, and many other steel structures are often found to be magnetized by the combined effects of the Earth's magnetic field and the vibrations created during construction. The effect is similar to stroking a needle with a strong magnet, in that the domains within the metals are caused to line up with the Earth's magnetic field.
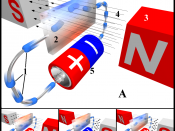
16.2 Electromagnetism
Moving electric charges produce a magnetic field.
Magnetic Field of a Straight Conductor
If a conductor is grasped in the right hand, with the thumb pointing in the direction of the current, the curled fingers will point in the direction of the magnetic field lines.
Magnetic Field of a Coil or Solenoid
If a solenoid is grasped in the right hand, with the fingers curled in the direction of the electric current, the thumb will point in the direction of the magnetic field lines in its core, and, hence, towards its north pole.
Using Electromagnets and Solenoids
In addition to its use in the well-known lifting electromagnets found in scrap-metal yeaards, it's used in bells, switches, relays, and magnetic speakers.
16.3 Magnetic Forces on Conductors and Moving Charges
If the right thumb points in the direction of the current, and the extended fingers point in the direction of the magnetic field, the force will be in the direction in which the right palm would push.
1 T is the magnetic field strength when a conductor with a current of 1A, and a length 1 m at an angle of 90 to the magnetic field, experiences a force of 1 N.
F = B I L sin 0
Force on a Moving Charge
F = Bqv sin 0
16.4 Ampere's Law
Along any closed path through a magnetic field, the sum of the products of the component of B parallel to the path segment with the length of the segment is directly proportional to the net electric current passing through the area enclosed by the path.
The Ampere As a Unit of Electric Current
1 A is the current flowing through each of two long, straight, parallel conductors 1 m apart in a vacuum, when the magnetic force between them is 2 x 10 N per metre of length.
1 C is the charge transported by a current of 1 A in a time of 1 s.
1 C = 1A * s
16.5 The Mass of the Electron and the Proton
e/m = 1.76 x 10 C/kg
16.6 The Oscilloscope
A device commonly used in the laboratory to analyze and measure electrical signals is the oscilloscope, whose major component is the cathode ray tube (CRT).
16.8 Electromagnetic Waves
Maxwell's Equations of Electromagnetism:
1. The distribution of an electric charge, in space, is related to the electric field it produces.
2. Magnetic field lines are continuos, and do not have a beginning or an end, whereas electric field lines
begin and end on electric charges.
3. An electric field can produce a magnetic field, so a changing electric field should produce a changing
magnetic field.
4. A changing magnetic field can produce a changing electric field, and hence an induced current and
potential difference in a conductor in the changing field.
Properties and Characteristics of Electromagnetic Waves:
1. Electromagnetic waves are produced whenever electric charges are accelerated. The accelerated charge loses energy that is carried away in the electromagnetic wave.
2. If the electric charge is accelerated in periodic motion, the frequency of the electromagnetic waves produced is exactly equal to the frequency of oscillation of the charge.
3. All electromagnetic waves travel through a vacuum at a common speed c, calculated at 3.0 x 10 m/s, and obey the wave equation c = f
4. Electromagnetic waves consist of oscillating electric and magnetic fields in a constant phase relation, perpendicular to each other, and both at 90 to the direction of propagation of the wave, as depicted in the sketch.
5. Electromagnetic waves exhibit the properties of interference, diffraction, polarization, and refraction, and can carry linear and angular momentum. Their intensity is proportional to the square of the magnitude of the electric field amplitude, and to the square of the frequency.
The Electromagnetic Spectrum
There is a broad range of frequencies of electromagnetic waves, called the 'electromagnetic spectrum', all having the same basic characteristics that Maxwell had predicted.
16.9 Applications of Electromagnetic Waves
There are many applications of electromagnetic waves in our 20th century, some of the most significant ones are described in detail below.
A. Radio and Television Communications
Marconi first recognized the potential for transmitting information over long distances using electromagnetic waves without any direct connection by wires.
Transmission
Sound waves are detected by a microphone and converted into a weak audio signal, containing frequencies in the range 20 Hz to 20 000 Hz. This signal is strengthened by an AF amplifier, then passes through modulator, where it interferes with an RF. This modulated signal, of either type, is then further amplified by an RF amplifier, and supplied to an antenna, where the complex mixture of frequencies and amplitudes is sent out in the form of an electromagnetic waves.
Reception
The first thing for the receiver is to select a certain carrier frequency to correspond to certain station. This can be accomplished by means of a resonant tuning circuit that contains an inductance (L) and a capacitance (C). This selected RF signal is then amplified and sent into a demodulator. Then AF signal is separated, and is amplified again, and then sent to the speaker for conversion into sound waves.
C. Blackbody Radiation
As the temperature of a solid increases, the radiation it emits is of higher and higher frequency.
Several key points about black body radiation:
÷ At a given temperature, a spectrum of different wavelengths is emitted, of varying intensity, but there is a definite intensity maximum at one particular wavelength
÷ As the temperature increases, the intensity maximum shifts to a lower wavelength.
D. Gamma Radiation
There three distinct types of radioactive materials:
1) a type of particle similar to high-speed helium nuclei, with poor penetrating power, called alpha particles.
2) a type of particle similar to high speed electrons, with the ability to penetrate up to several millimeters of aluminum, called beta particles .
3) a form of radiation, similar to very high energy X-rays, with the ability to penetrate even several centimetres of lead, called gamma rays .