Uranium, an element of the chromium group, never occurs naturally in a free state, although through processes of extraction it can be obtained from rare complex minerals such as pitchblende, carnotite, and uranite. Unfortunately, mining and using Uranium has become an issue that the world must tackle. At every stage of Uranium's life, problems arise and damage our world, whether it is in an environmental, social or ethical sense.
Before the uranium can be used it has to be mined. Pitchblende, a dark mineral that is the chief source of uranium, is "taken from the earth like any other metal, blasted and dug" from open-pit mining, surface mining and underground mining. Although, the creation of the mines causes great environmental damage because they not only destroy natural landscapes, they also drive people and other organisms from their habitats. Also the uranium mines and their processing operations in nuclear reactors produce sandy wastes called tailings.
These contain several radioactive elements, including thorium, radium, and radon, which emit low levels of radiation. This increases radiation risks to the workers, and environmental contamination.
Uranium needs to go through complex milling processes to extract it from pitchblende and other ores before it can be sold to nuclear reactors, in a form known as yellow cake (with the chemical symbol U3O8). First, pitchblende is broken up and is then exposed to strong acids such as sulfuric and nitric acids. Uranium dissolves to form uranyl sulfate, UO2SO4; others metals such as radium that also helped to form pitchblende ore are precipitated as sulfates. With the addition of sodium hydroxide, uranium is precipitated as sodium diuranate, also known as the yellow oxide of uranium, with the chemical formula: Na2U2O7 * 6H2O. Uranium can also be obtained from carnotite, and to do so the ore is...

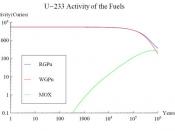
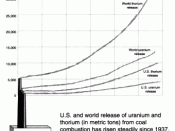
Too informal.
Don't take the first person in a formal research paper as this seems to be. Include a bibliography section as this information I assume didn't come fully formed onto paper without inspiration.
2 out of 2 people found this comment useful.