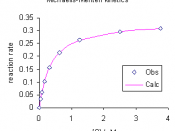
ABSTRACT: The purpose of the following laboratory experiment was to assess the kinetic behavior of metal ion-independent and CaN phosphatase activities in a soluble fraction from rat brain as a function of p-NPP concentration in relation to the Michaelis-Menten model for enzyme kinetics. A photometric procedure was used to quantify the amount of p-NPP substrate was consumed during the fixed-incubation period. The calculated results supported the hypothesis that both of these enzymes had obeyed the Michaelis-Menten kinetics. Both Michaelis-Menten plots were hyperbolic fits in which the initial reaction velocity is linear at low substrate concentrations and gradually reached a maximal level of saturation at higher substrate concentrations. The metal ion-independent phosphatase with Vmax and Km values of 28.17 nmol/mg/min and 21.78 mM, respectively had exhibited a difference in the ability to use p-NPP as a substrate by comparison to these values for CaN phosphatases at 25.64 nmol/mg/min and 2.856 mM, respectively.
It was also denoted that in both cases at p-NPP concentrations greater than 20 mM there was a slight decrease in the saturated portion of Michaelis-Menten plot, indicative of the presence of inhibition within the system.
INTRODUCTION: Almost all of the reactions that occur within a cell are catalyzed by enzymes. While the specific substrates and products of these reactions vary greatly, the basic properties of all enzyme-catalyzed reactions are similar. Enzymes are catalysts. That is, they help a reaction proceed without being altered. The substrate binds to a specific site on the enzyme, and is converted to the product(s) and then released. Without the presence of the enzyme, the reaction would occur, but at a dramatically slower rate.
In biochemical reactions, the catalytic event that converts substrate to product involves the formation of a transition state, and it occurs most easily at a specific binding site on the...