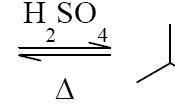ABSTRACT
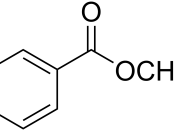
This experiment was conducted to synthesize methyl benzoate form benzoic acid and methanol by using the Fischer esterification method. The Fischer exterification technique is utilized in the academic and industrial settings due to the simplified synthesis and safety parameters of the overall reaction. Both benzoic acid and methanol are relatively cheap to obtain from a commercial source as well as being easy to store with a relatively long shelf life. The Fischer esterification method is a fundamental and important synthesis process in training our future chemists in both commercial and academic employment and principles. By utilizing this method of synthesis, the chemist has the knowledge to utilize certain chemicals to facilitate further reactions and derive pertinent data of said reactions by utilizing differing laboratory equipment, i.e., nuclear magnetic resonance (NMR), mass spectroscopy (MS), infrared (IR), and gas chromatography (GC) for analysis. These techniques allow the chemist to derive functional groups and isolate unknown compounds that can verify and identify unknown compounds often obtained through academic and commercial laboratory research projects.
INTRODUCTION
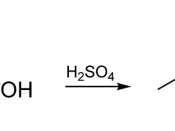
The actual conversion of carboxylic acid to esters using an acid catalyst and an alcohol was given the name Fischer esterification after the German chemist, Emil Fischer (1). The basic mechanism for this reaction is shown below in Fig. 1 (5). The reaction begins with the transfer of a proton from an acid catalyst to an alcohol molecule. A proton transfer follows from the protonated alcohol and finally to the carboxylic acid. The protonated carbonyl of the acid is attacked by the lone pair of electrons on the hydrogen atom of the alcohol for the oxonium ion. A proton transfer follows from the oxonium ion to an alcohol molecule, generating a tetrahedral carbonyl addition intermediate. One of the OH groups I the intermediate abstracts a proton from the protonated methanol molecule to form a very good leaving group, which is water. In the final step, the methanol molecule abstracts a proton form the protonated carbonyl to regenerate the carbonyl and whether molecule is displaced. The final product is the ester.
Fig. 1 Mechanism of Reaction (5)
Fischer esterification is an equilibrium reaction (2). The rate or equilibrium constant K is dependent upon the simple formula of products over the reactants and can be manipulated by the basic principles of Le Chatlier (2). One can increase or decrease the rate of the reaction by using these basic principles. One can increase the cheaper reactant such as an alcohol (methanol in our reaction), remove a product, such as water, using the Dean Stark apparatus, or one can manipulate the pressure or temperature. Most organic chemists do not get involved with pressure manipulation; the results can often be dangerous when conducting experiments utilizing high pressures. Manipulating the reaction using these different principles will allow the chemist to yield more of the ester compound by maximizing the reaction potential. The fact that the reaction is an equilibrium reaction opens the door for side product such as reactants being isolated in the final product as well. The reaction has the ability to go both ways.
MATERIALS AND METHODS
Place 2.0 g of benzoic acid and 5.0 ml of methanol in a 10.0 ml conical vial (2). Cool this mixture in an ice bath for approximately 10 minutes; then add 0.6 ml of concentrated sulfuric acid. Swirl the vial to mix the components of the mixture (2). Next, add a boiling stone and assemble a reflux apparatus using a water-cooled condenser.
Procure a hot plate and sand bath to bring the mixture to a boil. Make sure to reflux the mixture for at least one hour. Remove from sand bath and allow cooling to room temperature. Decant the mixture into a small separatory funnel containing 10 ml of water and rinse the vial with 10 ml diethyl ether (2). Add to the separatory funnel and mix vigorously. Drain off bottom (water) layer. This layer will contain the sulfuric acid and excess methanol. Wash the remaining ether layer in the funnel with 6.0 ml of water followed by 6.0 ml of 0.5 M sodium bicarbonate to remove all the unreacted carboxylic acid (2). Shake well and vent frequently to release pressure. Repeat the bicarbonate washing process until the solution is basic when testing with pH paper. Drain the bicarbonate layer into a 50.0 ml beaker, then wash the ether layer with a few milliliters of saturated sodium chloride solution, then dry the solution over calcium chloride pellets. Make sure to add calcium chloride pellets until no clumping is observed.
After approximately 10 minutes, decant the ether into a 125.0 ml vacuum flask. Add approximately 3 - 4 ml of fresh ether to rinse the drying agent (2). After transferring the product to the flask, cap it by using an adaptor and evaporate the solvent using an aspirator. Once the solvent is evaporated, oil will remain. This oil will need to be purified by distillation. Set up the Hickman Still apparatus and air condenser and place the apparatus in a sand bath to heat the oil until it boils (2). The boiling point of ester is approximately 199 degrees Celsius; collect all material boiling above that temperature (2). Pre-weigh a conical vial and transfer the distillate. After weighing the product, calculate the percent yield and take the product to the IR to obtain a spectrum of the pure product (2).
DATA AND RESULTS
As a result of the Fischer esterification process, 0.344 g of methyl benzoate was recovered for the final product. The calculation for the yield obtained by the synthesis is provided below (3):
0.344g
% Yield =
% Yield =
DISCUSSION
The synthesizing of methyl benzoate from benzoic acid and methanol was a successful endeavor. This reaction actually produced the product better than anticipated in a laboratory setting. Based on the I.R. spectra obtained; we can conclude that we had formed the product. The first peak was in the 3066 cm-1 range being the C=C-H, and the C-H at the 2953 cm-1 peak (1). We also have the C=O peak at 1725 cm-1 along with the C=C at 1602 and C-O-C at the 1279 cm-1 range (2). This structure verifies our final product of methyl benzoate. We recovered 0.344 g of the final product (methyl benzoate) for a 15.46% final yield. Methyl benzoate has a boiling point of 199.5 degrees Celsius and a molecular weight of 136.15 grams per mole (3). With a boiling point this high, it was important to reflux for the entire hour. We refluxed for just over one hour and then allowed it to cool to room temperature. At this point we used the separatory funnel to separate our products and wash, then dry by evaporating the liquid on the hot plate.
REFERENCES
1.Fischer, E. Available at http://www.nobel.se/chemistry/laureates/1902/fischer-bio.html. Downloaded 28 Wednesday 2004.
2.Salvatore RN, Jung KW. experimental organic chemistry: A synthetic & mechanistic perspective. 222 Berkley St. Boston: Houghton Mifflin; 1999. 102 p.
3.Density and formula weight of methanol. Available at http://chemfinder.canbridgesoft.com/result.asp. Downloaded 28 Wednesday 2004.
4.Synthesis of methyl benzoate. Available at http://www.mccfl.edu/Faculty/GambinoA/CHM%202211/Labs%20in%20Word/Synthesis%20of%20Methyl%20Benzoate.DOC. Downloaded 28 Wednesday 2004.
5.Mechanism of reaction. Available at http://www.chem.ubc.ca/courseware/260/Reactions_Acids_and_amides.pdf. Downloaded 28 Wednesday 2004.