ABSTRACT
For hundreds of years, spices have been used by sailors to inhibit the fermentation of the
alcohol they transported. These spices, including Clove were used with good reason. In
this study it was found that 1% clove solution inhibited a fermentation reaction by as
much as 0.093ml/min. The clove solution also decreased the total evolution of CO2 by as
much as 2.845 ml.
The process of fermentation is better inhibited using microbial inhibitors such as clove,
than the natural inhibitor ethanol.
INTRODUCTION
Microorganisms are capable of an amazing array of different types of fermentation. Most
natural compounds are degraded by some type of microbe and even many man-made
compounds can be attacked by bacteria. In environments devoid of oxygen (or other
suitable inorganic electron accepter), this degradation involves fermentation.
Despite the many methods bacteria employ to ferment organic compounds, there are
some unifying concepts that are true of all fermentations.
1.) NAD+ is almost always reduced to NADH
Metabolism involves the oxidation of the substrate. These electrons are removed from the
organic molecule and most often given to NAD.
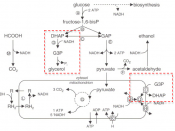
Figure 1 - oxidation of glyceraldehyde-3-phosphate to 1,3 bisphosphoglycerate. Electrons are removed from the carbon denoted in red and donated to NAD+. An inorganic phosphate is attached to the carbon shown.
2.) Fermentation results in a excess of NADH
Accumulation of NADH causes a problem for anaerobes. They have too much of it and it
prevents further oxidation of substrate due to a lack of an NAD+ pool to accept electrons.
In many fermentation pathways, the steps after energy generation are performed in part to
get rid of the NADH.
Pyruvate is a valuable intermediate because it can be used for cell synthesis and many
different enzymes can act on it. It gives the microbe flexibility.
3.) Energy is...