Give an account on the molecular structure of carbohydrates
Carbohydrates contain the elements of carbon, hydrogen and oxygen. Examples of carbohydrates include sugars and starches. There are three main types based on size.
Monosaccharides
Monosaccharides contain a single sugar molecule. The general formula is (CH2O)n, where 'n' equals the number of carbon atoms in the molecule. Different monosaccharides have different numbers of carbons. Trioses have 3 carbons, Pentoses contain 5 and Hexoses contain 6 carbons.
Examples of monosaccharides include alpha glucose and beta glucose. In alpha glucose the hydroxyl group is found below carbon 1, whereas in beta glucose the hydroxyl group in found above. Glucose is the most important monosaccharide. It has a 6-carbon hexose sugar. In animals it is the main transport sugar in the blood, and its concentration in the blood is carefully controlled.
There are many isomers of glucose such as galactose and fructose. They have the same chemical formula (C6H12O6) but different structural formulae.
Disaccharides
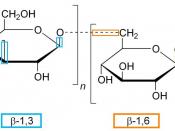
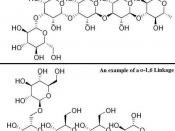
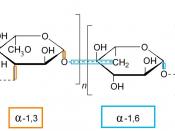
Disaccharides are a double sugar unit made up of 2 monosaccharides, which are joined by a glycosidic bond during a condensation reaction.
Maltose has an alpha 1-4 glycosidc bond. This means that water is formed as a product during the reaction from carbon 1 from 1 alpha glucose and carbon 4 on another. Maltose is found in germinating barley products of starch digestion with amylase.
Other examples include sucrose, which is made up of glucose and fructose. And lactose which has glucose and galactose monomers present. The chemical formulae of disaccharides is C12H2O11 since H2O was removed during the condensation reaction.
Polysaccharides
Polysaccharides are made up of many sugar monomers joined by glycosidic bonds. They may be straight, branched or helical. They can be very large molecules such as starch, glycogen and cellulose.
Starch is a plant storage carbohydrate with...