Chemistry 121 April 26, 2001 r What is the Ozone Hole and What Are Its Causes? Abstract: Ozone depletion is one of many environmental problems burdening society today. I will describe to you the chemistry involved in making ozone and how ozone depletion occurs. I will tell you why I decided to conduct research on this issue. I will tell the methods of research I used to gather information on ozone depletion. I will state my position on the debate of the ozone hole and propose an experiment to bring society one step closer to an ending of the issue.
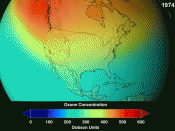
Introduction: Ozone is a compound composed of three oxygen atoms (O3). CFCs (Chlorofluorocarbons), are harmful compounds that are released into the air mainly by industrial factories. When the CFCs reach the stratosphere, UV rays produced by the sun strike the compound causing the chlorine atom to break away from the compound (2) .
The free chlorine atom will then search for an ozone molecule and collide with it(2). The chlorine atom will steal an oxygen atom from the ozone molecule, thus destroying the ozone molecule(2). Two new compounds are formed, Chlorine-Oxide (ClO) and Oxygen (O2). The newly formed chlorine-oxygen compound will eventually make a collision with a free oxygen atom [Cl + O3 ÃÂî ClO + O2]. (O2) will form and the chlorine atom is now free to attack other ozone molecules and repeat the ozone depletion process(2).
Rationale: I decided to research the ozone hole because it was the most influential topic out of three in my life. The car I drive does not have an air bag so there is no direct influence on my life from it. Global warming is an important issue, but the process is so long that by the time the temperature of earth...