Outline
- Introduction
- Structure of carbohydrates
- Functions of carbohydrates
- Tests for carbohydrates
I. INTRODUCTION
- Carbohydrates are substances that contain the elements carbon, hydrogen and oxygen.
- They have the general formula Cx(H2O)y, where x and y are variable numbers.
II. STRUCTURE OF CARBOHYDRATES
- Carbohydrates are divided into three main classes:
- Monosaccharides
- Disaccharides
- Polysaccharides
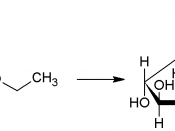
1. Monosaccharides
- Defined as a carbohydrate which cannot be hydrolysed to simpler carbohydrate.
- The general formula is (CH2O)n
- Common examples are glucose, fructose and galactose (hexose sugars)
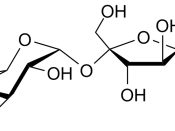
2. Disaccharides
- A disaccharide is formed by a condensation reaction between two monosaccharides - one molecule of water is removed from the pair of monosaccharides (Fig. 1).
Common examples are:
- maltose = glucose + glucose
- lactose = glucose + galactose
- sucrose = glucose + fructose
- The bond formed between the 2 monosaccharides is called the glycosidic bond.
1
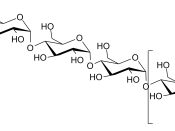
3. Polysaccharides
- Polysaccharides are polymers of monosaccharides.
Common examples are:
- Starch (storage polysaccharide)
- Glycogen (storage polysaccharide)
- Cellulose (structural polysaccharide)
* Structure of starch
- Starch is a polymer of glucose
- It has two components: amylose and amylopectin.
i. Amylose
- It has a straight chain structure.
- Consists of several thousand glucose residues joined by ñ(1-4) glycosidic bonds.
- These bonds cause the chain to join helically into a more compact shape
ii. Amylopectin
- It is also compact but has a highly branched structure.
- The branches are formed by ñ(1-6) glycosidic bonds.
- It has twice as many glucose residues as amylose
III. FUNCTIONS OF CARBOHYDRATES
1. Monosaccharides are for synthesis of nucleic acids (ribose is a constituent of RNA, deoxyribose is a constituent of DNA) and synthesis of disaccharides and polysaccharides.
2. As a source of...