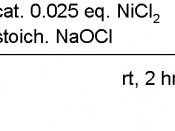
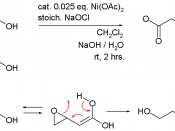
Name: nickel
Symbol: Ni
Atomic number: 28
Atomic weight: 58.6934 (2)
Minerals containing nickel were of value for colouring glass green. The mineral used for colouring glass was called kupfernickel (false copper). Nickel was discovered by Baron Axel Frederik Cronstedt in 1751 in a mineral called niccolite. Apparently, he had expected to extract copper from this mineral but got none at all, obtaining instead a white metal that he called nickel after the mineral from which it was extracted.
Discovered by:
Axel Fredrik Cronstedt
Discovered at:
Sweden
Discovered when:
1751
Origin of name:
From the German word "kupfernickel" meaning Devil's copper or St Nicholas's (OLd Nick's) copper
Fluorides
NiF2: nickel (II) fluoride
Chlorides
NiCl2.6H2O: nickel (II) chloride 6-water
NiCl2: nickel (II) chloride
Bromides
NiBr2: nickel (II) bromide
Iodides
NiI2: nickel (II) iodide
Oxides
NiO: nickel (II) oxide
Ni2O3: nickel (III) oxide
Sulfides
NiS: nickel (II) sulphide
NiS2: nickel disulphide
Ni3S2: nickel sulphide
Selenides
NiSe: nickel (II) selenide
Ground state electron configuration: [Ar].3d8.4s2
Shell structure: 2.8.16.2
Term symbol: 3F4
Melting point [/K]: 1728 [or 1455 ðC (2651 ðF)]
Boiling point [/K]: 3186 [or 2913 ðC (5275 ðF)] (liquid range: 1458 K)
Density of solid [/kg m-3]: 8908
Year Scientist(s) Discovery
Greek
era Democritus
"by convention bitter, by convention sweet, but in reality atoms and void"
1704 Isaac Newton
Proposed a mechanical universe with small solid masses in motion.
1803 John Dalton
Proposed an "atomic theory" with spherical solid atoms based upon measurable properties of mass.
1832 Michael Faraday
Studied the effect of electricity on solutions, coined term "electrolysis" as a splitting of molecules with electricity, developed laws of electrolysis. Faraday himself was not a proponent of atomism.
1859 J. Plucker Built one of the first gas discharge tubes ("cathode ray tube").
1869 Dmitri Mendeleev
Arranged elements into 7 groups...