Hydrogen's Dirty Secret
Although hydrogen fuel-cell cars have their advantages to petrol-fueled internal-combustion automobiles, they still don't offer the performance nor the convenience that most consumers desire. As of now, automotive engineers still have a long way to go before they could compete with gas-operated automobiles. There are plenty of obstacles that scientists have to overcome in order to make the hydrogen fuel-cell car up to par with the petrol-fueled car.
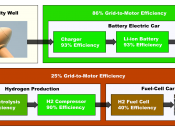
There are plenty of people who promote hydrogen-run cars. Their main selling point is the fact that the only emission given off by these cars are heat and water vapor. Although this is true, if one were to research farther, they would realize an important point pro-hydrogen enthusiast failed to mention. Although hydrogen is in fact plentiful, there are very little or almost no free-floating hydrogen molecules on Earth. We know, of course, that most hydrogen molecules are bound to oxygen molecules which, together, make water; but there is no other source that we know of that is as plentiful in hydrogen as water is.
Therefore, if we want to use the hydrogen in water, we must find a way to separate the hydrogen molecules from the oxygen molecules. This presents us with a problem.
Where would the energy to break the hydrogen bonds come from? The main choices are either fossil fuels or nuclear power, both of which are major not possible because that would create for the environmental issues these pro-hydrogen enthusiasts sought to avoid in the first place. One could also suggest the use of some sort of plants that create hydrogen, but then there would still be greenhouse gas emissions to associate with cars present, even if it isn't from the cars themselves.
But let us imagine we did find a way to make the...


Good work
Nice points, great structure: you just need a summary at the end to conclude the essay.
7 out of 7 people found this comment useful.