SCIENCE COURSEWORK
Aim: To investigate how concentration effects osmosis on potato
chips.
Introduction:
I have been asked to investigate the effect of osmosis on potato chips. Osmosis is the diffusion of water from a lower concentration of solute to a higher concentration of solute, through a membrane.
I have chosen to investigate the effect of the water potential/concentration of solution on potato chips. This means that I will put potato chips into sucrose solutions of different concentrations. These concentrations would be ones such as these; 0molar, 0.2m, 0.4m, 0.6m, 0.8m, 1.0 and distilled water.
There are other factors involved and these are;
1) Temperature
2) Water potential of potato initially
3) Size of potato
4) Light intensity
5) Mass of potato dependent variable
6) Volume of solution potato chip is in
7) Type of potato
8) Time left in solution
9) Surface area of potato chip
10)Use the same balance to measure chip
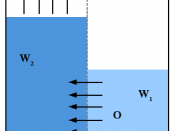
Osmosis is the movement of water down a concentration gradient through a partially permeable membrane.
Osmosis through a partially permeable membrane continues until the concentration of water on either side of a membrane is equal.
Osmosis is a special kind of diffusion; it happens when a membrane has tiny holes in it, which let water molecules through but stop larger molecules, like sugar. A membrane like this is called semi-permeable.
This experiment shows osmosis: -
Cell A Cell B
because osmosis is the movement of particles from a weak
concentration to a strong concentration through a semi permeable membrane the water will move from the beaker into the visking tubing.
Partially
Permeable
Membrane.
Before After
Osmosis Osmosis
Water molecules
Partially Weak strong
Permeable Solution solution
membrane
Water molecules diffuse from a high water concentration to a lower water concentration.
A wilted leaf will be more concentrated because...