AbstractThis investigation is a study on the distribution of catalyse in different plant tissues. I looked at 7 different tissues and measured the amount of oxygen given off to see the amount of catalyse present. I found varying tissues to have varying amounts present.
IntroductionCatalase is a widespread enzyme, found in nearly all aerobic cells (animals, plants and microbes). It protects the cell from the toxic effects of hydrogen peroxide, generated as a waste by-product of cell metabolism. It does this by catalysing the decomposition of hydrogen peroxide (a powerful and potentially harmful oxidizing agent) into molecular oxygen and water. The reaction can be summarised by the equation:2H 0 2H 0 + 0Different plant materials contain very different amounts of catalase activity and this is what I will be looking at.
Catalase is located in a cell organelle called the peroxisome. Peroxisomes in plant cells are involved in photorespiration (the use of oxygen and production of carbon dioxide) and symbiotic nitrogen fixation (the breaking apart of the nitrogen molecule to reactive nitrogen atoms).
Hydrogen peroxide is produced during these chemical processes and must be removed to prevent damage to cellular structures. If hydrogen peroxide is added to tissue containing the enzyme catalyse bubbles are produced, this is evidence of oxygen production and confirms that there was catalyse present.
Catalase has one of the highest turnover rates for all enzymes: one molecule of catalase can convert 6 million molecules of hydrogen peroxide to water and oxygen each minute.
AimsTo investigate the amount of catalyse in varying plant tissuesHypothesisThe more a plant is respiring, the higher the catalyse activity and the more oxygen will be produced.
VariablesThere are many variables, which could affect my results these are:â¢Temperature- the higher the temperature, the higher the rate of reaction up to a certain point. Whilst the optimum temperature of catalyse is probably higher than room temperature, about 20 degree Celsius, I shall and keep my experiment reacting at room temperature which is about 22 degrees Celsius.
â¢Surface area-the bigger the surface area the quicker the reaction will occur as the hydrogen peroxide reacts with the catalyse. I shall over come this by cutting all the tissues into 3 mm cubes.
â¢Time-I will leave the experiment for 5 minutes in order to give enough time for the reaction to occur.
â¢The concentration of Hydrogen Peroxide-In order to make the experiment a fair test I shall use 10 volume of the hydrogen peroxide.
â¢Amount of tissue-the amount of tissue will help determine the amount of catalyse that will be present, which in turn will affect the amount of oxygen given off. In order to keep the fair test I will use the same weights (3grams) each time.
SafetyWhen working with the hydrogen peroxide I have to be extremely careful as it can cause burns to skin or clothing as it is a corrosive. I will have to make sure that if I spill any or get any on me I wash it off using plenty of water and if severe seek medical attention. When working with it I shall use eye protection and gloves.
PilotIn order to make sure my equipment and experiment works I am going to trial it.
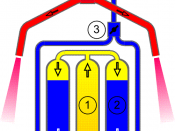
The tissues I shall use will be:â¢Appleâ¢Potatoâ¢Carrotâ¢TurnipApparatusI will be using:â¢A 1 cm syringeâ¢Glass delivery tubeâ¢Stopwatchâ¢Boiling tubeâ¢Clampâ¢Litre beakerâ¢Inverted barrel of a 20 cm syringeâ¢Chopping boardâ¢Knifeâ¢Weighing scalesMy method is generally adding hydrogen to different plant tissues which may or may not contain varying amounts of catalyse, I will measure the amount of catalyse present by the amount of oxygen produced. Below you can see a diagram of how it was set up.
Pilot Method1)Prepare all tissues by chopping them up into 1cmó squares and weighing them out to weigh 3grams. I decided on 3 grams as it wasn't too little to get a reaction and wasn't too much for the hydrogen peroxide to react with.
2)Set up my apparatus as shown in diagram above, using clamps to support the boiling tube and the rubber tubing attached to the barrel of the 20 cmó syringe.
3)Remove the 1cmó plastic syringes, and then remove the bung from the boiling tube.
4)Insert 3 grams of the tissue into the boiling tube and replace the bung.
5)Open the screw clip to draw water into the barrel of the 20cmó syringe, close the clip when the barrel is full.
6)Draw up 1cm of hydrogen peroxide into the syringe and then insert it into the boiling tube.
7)Depress the plunger of the 1cmó syringe to inject the hydrogen peroxide solution into the boiling tube.
8)Start the stopwatch, measure and record the volume of oxygen collected in the barrel of the 20cmó syringe over a period of five minutes.
Pilot resultsTissueAmount of oxygen produced (ml)Apple0Potato1Carrot0Turnip0Due to my pilot experiment not working as well as I had planned and hoped, I have made some changes to the method and have added more varying tissues.
I decided that I would use:â¢Developing Bluebellâ¢New growing leavesâ¢Germinating broad beansI decided to do this so that I would get results of tissues that should be respiring a lot.
I also decided that in order to increase the surface area of the tissues I would blend it up in a blender. In order to minimise large chucks I had to add 10ml of water to make a solution, this then took the large chucks off the outside of the blender and thus made them get caught up in the blades, thereby pulping the remaining chucks of tissues completely. I did this for all of my tissues and mixed it for about 20 seconds each.
As I got no reaction I decided to add more hydrogen peroxide, I increased it from 1cm to 2 cm .
In order to try and keep the test tube with catalyse in it reacting at room temperature I placed it in a water bath. This will help keep it a fair test and will stop any fluctuations in room temperature affecting my results.
Everything else about the pilot experiment I did the same.
Results123456AveragePotato1110998109.5Carrot10547656.1Bluebells5645475.1Beans4568475.6Apple0010000.16Leafs2531242.8Turnip3455654.6AnalysisWith reference to my graph above, I can see that generally most plant tissues contain some catalyse, however apple seems to show signs of having none or very little and potato having the most.
Storage organs such as the potato are high in catalase activity as proven by my experiment as they gave off the most oxygen gas. Also the tissues which are respiring a lot such as the germinating beans and bluebell buds are high.
My results are fairly accurate as they have been in a lab controlled situation as so all variables are managed and so should be more accurate.
My results show to me the apple does not have any catalyse activity present in its tissues; this means no respiration is occurring due to the plant not having to protect itself from the harmful effects of hydrogen peroxide.
ConclusionCatalyse activity is most present in tissues which are respiring quickly such as new plant tissue or tissues which are being held as storage such as the potato. My experiment has proven that certain plant tissues do have more catalyse activity then others.
EvaluationMy accuracy of my investigation has to be criticised to analyze. I think there were a couple of problems with my results. Firstly there may have been errors in my equipment such as the bung and test tube may have been not tightly fitted enough and so my results would produce a systematic error producing the same biased result each time but it would have still been a fair test.
The experiment produced me the exact measurements I wanted and generally I produced the results that I was expecting.
There were errors in my experiment the main one was how enough oxygen had to be produced in order to even get a result as the oxygen had to be sufficient to travel round the delivery tube and into the up turned syringe, this would be a problem on tissues with a small amount of catalyse activity as I would think it didn't react at all due to no oxygen reaching the inverted syringe, however on all the tissues that did react a systematic error would be produced as it would take the same amount of oxygen each time to reach the syringe.
Overall I think my investigation was successful as I proved my hypothesis to be correct and I got overall the results I would have predicted.
BibliographyEncyclopaedia Britannicawww.catalase.com/catalinks.htm