Prediction
Osmosis is defined as 'the movement of water molecules from an area of high water concentration to an area of low water concentration, across a semi-permeable membrane' (Collins, 1999).
If you get information from books put the copied text in brackets and at the end put the authors surname and date the book was published in brackets. Then put the full details of the book at the end of the course work in the 'References'
In a high concentration of water the amount of solute (e.g. sugar) is low. This could be called a weak or dilute solution.
In a low concentration of water the amount of solute (e.g. sugar) is high. This could be called a strong or concentrated solution.
When two such solutions are divided by a semi-permeable membrane the water will move from the area of high concentration to the area of low concentration, until both sides are equal (have reached equilibrium).
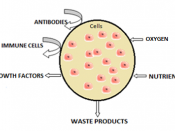
This can be seen in living cells. The cell membrane in cells is semi-permeable and the vacuole contains a sugar/salt solution. So when a cell is placed in distilled water (high water concentration) water will move across the semi-permeable membrane into the cell (lower water concentration) by osmosis, making the cell swell. This cell is now referred to as turgid. If done with potato cells the cells would increase in length volume and mass because of the extra water.
If these potato cells were placed in a solution with a low water concentration, then the opposite would happen. Water would move out of the cell into the solution. In extreme cases the cell membrane breaks away from the cell wall and the cell is referred to as plasmolysed. The potato cells will have decreased in length, volume and mass.
The greater the concentration...

Dalbir is the best
great work again from dalbir
2 out of 2 people found this comment useful.