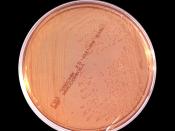
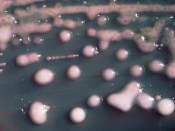
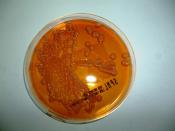
Characteristics of Gram Negative Organisms
What is in the water? Beyond the animals and plants we can see with the naked eye, there are several species of microorganisms that inhabit natural water sources. These microorganisms are important to help sustain a healthy water environment. However, some of these microbes can cause harm to the organisms that also live within or use the water as a drinking source. Therefore, it is important to identify microorganisms within the water to avoid disease. Common types of gram negative bacteria that inhabit water are from the families of Enterobacteriaceae, Vibrionaceae, Pseudomonadaceae, Neisseriaceae and Pasteurellaceae. This laboratory experiment will be used to identify a gram negative microbe but do not forget that there are several types of gram positive bacteria that live in the water too!
1) ISOLATION OF GRAM NEGATIVE ORGANISM BY SPREAD PLATE PROCEDURE
Label MacConkey agar plate. This media will select for gram negative organisms.
Using a fresh pipette transfer 0.1ml of the water to the surface of an agar plate.
Spread the organisms over the surface of the agar using a glass spreader.
Sterilize the glass spreader by dipping in alcohol and passing through the Bunsen flame. DO NOT hold the spreader in the flame. Hold the spreader away from the flame while the alcohol burns off.
After the spreader has cooled, you may touch it to the inside surface of the petri dish lid to ensure it is cool, spread the bacterial sample over the surface of the medium by rotating the plate on a turntable.
When you have finished, flame the spreader, to ensure it is sterile, before putting it down on the bench.
Incubate the plates at 37oC for 24hrs.
Select one colony from the MacConkey agar and subculture it onto TSA. Note its characteristics on the MacConkey agar.
2) MACCONKEY AGAR
MacConkey agar is a selective and differential medium that permits the recovery and selection of enteric organisms (Enterobacteriaceae). Bile salts and crystal violet are included in the medium to inhibit the growth of Gram positive and some fastidious Gram negative organisms. Lactose is present as the sole carbohydrate, with neutral red added as a pH indicator to detect fermentation of the sugar. Neutral red is colorless at pH>6.8 and red at pH<6.8. Organisms fermenting lactose result in production of acid which turns the indicator red. Lactose fermenters therefore turn red. As acid accumulates it also precipitates the bile salts with absorption of the neutral red, resulting in the medium surrounding the growth also turning red. Non-lactose fermenting organisms do not produce acid so the colonies are uncolored or transparent.
Procedure
Divide a MacConkey plate into sections by marking the bottom of the petri dish.
On separate sections inoculate your bacterial cultures.
Incubate the plates at 37oC for 18-24 hours.
3) CYTOCHROME OXIDASE
Cytochrome oxidase (aka indophenol oxidase) is involved in the terminal step in the electron transport chain which catalyzes the oxidation of a reduced cytochrome (cytochrome c) by oxygen. When assaying for oxidase do not take cultures from media containing glucose; fermentation inhibits the activity of cytochrome oxidase which may then yield false negative results. In the laboratory cytochrome oxidase can be determined by a color reaction in which a blue product is formed. The reaction is:
N,N,N',N' tetramethyl--phenylenediamine dihydrochloride + oxygen indophenol + H2O
Procedure
Hold reagent dropper upright, break ampule by squeezing between thumb and forefinger. Tap on table top, then invert for drop-by-drop dispensing
Add a few drops of oxidase test reagent to piece of filter paper. Do not use excess reagent as it may cause reaction to fade on oxidase-positive organisms.
Using wooden applicator stick pick up some growth and rub gently on to area of reagent
Observe the color of the inoculated area: violet-purple color is a positive reaction, a negative reaction is the absence of a blue color.
Allow to stand for no longer than 30 seconds. Delayed reactions should be ignored.
Discard the oxidase test strips into the Biohazard containers.
*****NOTE*****
BASED ON OXIDASE RESULTS WILL DETERMINE NEXT SERIES OF BIOCHEMICAL TESTS
4) IMViC TEST SERIES
Members of the Enterobacteriaceae (found in intestinal tract) are short gram negative, non-spore forming rods; they include pathogens (Salmonella sp.), opportunistic or occasional pathogens (Proteus sp.) and non-pathogens (Escherichia sp.). Differentiation between members of the Enterobacteriaceae is by their biochemical properties; one series of tests known as the IMViC series is extremely useful in the differentiation. The IMViC series of tests comprise the indole, methyl-red, Voges-Proskauer, and citrate utilization.
a) INDOLE PRODUCTION
Indole is a metabolic product of the degradation of tryptophan. Hydrolysis and deamination of tryptophan, by tryptophanase, results in the production of indole, pyruvic acid and ammonia.
CH2 CH COOH CH3
NH2 + C=O + NH3
N N
H H COOH
tryptophan indole pyruvic acid ammonia
Indole is detected by reaction with the aldehyde group of -dimethylamino-benzaldehyde. Kovacs reagent contain -dimethylaminobenzaldehyde, butanol and hydrochloric acid. The acidified butanol extracts any indole produced in the medium into the reagent layer where it can react with -dimethylamino-benzaldehyde resulting in the formation of a cherry red color.
Procedure
Inoculate tryptophan broth.
Incubate at 37oC for 24-48 hours.
Add 10-15 drops of Kovac's reagent. Run the reagent down the inner wall of the tube such that the reagent forms a layer on top of the medium.
Development of a cherry red color (within 10-15 seconds) at the interface of the reagent and the broth indicates the presence of indole.
b) METHYL RED TEST
Fermentation of glucose can occur via a number of pathways, one of which, the mixed acid fermentation pathway, results in the formation of a very low pH due to production of strong acids. Glucose fermentation via the mixed acid fermentation results in the formation of stable acid end products such as acetic, lactic, succinic and formic acids. Because many Enterobacteriaceae produce acids during the early stages (18-24 hours) of growth which are later converted to more neutral compounds it is important to run the test for at least 48 hours, although up to 5 days incubation can be used.
Production of strong acids are detected by addition of methyl red which has a pH range between 6.0 (yellow) and 4.4 (red). To maintain the red color on addition of methyl red, large quantities of strong, stable acids must be produced from the carbohydrate source. Organisms not producing stable acids result in formation of a change in color of the pH indicator to yellow.
Procedure
Inoculate 3 ml of MR/VP broth medium.
Incubate at 37oC for 48-96 hours.
Add 5 drops of methyl red pH indicator.
Development of a stable red color at the surface of the medium indicates the presence of sufficient acids to lower the pH to 4.4. This is a positive methyl red (mixed acid fermentation) test.
c) VOGES-PROSKAUER TEST
Some organisms ferment pyruvate resulting in production of 2,3 butanediol:
glucose + O2 pyruvic acid acetoin 2,3-butanediol + CO2 + H2
An intermediate in this pathway is the compound acetoin (acetylmethyl-carbinol). Organisms that utilize this pathway produce smaller amounts of mixed acids, generally insufficient to result in a positive methyl red test.
2,3-Butanediol is not a very reactive compound so in the VP test the presence of the intermediate acetoin is assayed. In the presence of atmospheric oxygen and potassium hydroxide, acetoin is converted to diacetyl, with -naphthol serving as a catalyst, resulting in formation of a pink-red color change. It is important that this test be read after 24-48 hours. Short incubation times will not allow for sufficient time to accumulate acetoin, whereas after prolonged incubation acetoin will be converted to 2,3-butanediol.
Procedure
Inoculate 3 ml of MR/VP broth medium.
Incubate at 37oC for 24-48 hours.
Add 30 drops of -naphthol. Mix thoroughly.
Add 10 drops of 40% KOH. Mix well.
NOTE: it is important that the reagents get added in this order.
Incubate the tubes in a slanted position (increases surface area) for up to 60 minutes. Do not exceed 60 minutes, as a false positive test may be obtained.
Development of a pink-rose color is a positive VP test. A copper-like color, formed by reaction of KOH with -naphthol should not be mistaken for a positive test. This reaction may occur on prolonged incubation following addition of the reagents.
d) CITRATE UTILIZATION
The citrate utilization test is used to differentiate between microorganisms that are able to use citrate as their sole carbon source. Sodium citrate is one of the metabolites of the TCA cycle, and some bacteria are able to utilize this to obtain energy by means other than fermentation. Utilization of citrate is detected by an increase in the pH of the medium (production of alkaline products, degradation of acidic functional groups). Simmon's citrate agar is a defined medium with sodium citrate as the sole carbon source and ammonium ions as the sole nitrogen source. The agar is poured as a slant to increase the surface area as formation of the alkali products is an aerobic process. Bromothymol blue is incorporated as a pH indicator, being yellow at pH<6.0 and blue at pH>7.6. The initial pH of the medium is around pH 6.9, at which the bromothymol blue is green.
Procedure
Inoculate a Simmons Citrate agar slant. Use only a light inoculum as a heavy inoculum will carry over preformed organic compounds that the cell could utilize.
Incubate at 37oC for 24-48 hours.
A positive test is indicated by the development of a blue color in the medium.
5) THIOSULFATE CITRATE BILE SUCROSE AGAR (TCBS)
TCBS is selective and deferential medium used to promote growth of Vibrio species because of the alkaline pH (8.6). This agar contains Oxgall and sodium cholate that inhibits the growth of most gram positive microorganisms (i.e. selective medium). TCBS is used to differentiate between fermenters and non-fermenters of sucrose. A positive sucrose fermenter will appear as a yellow colony on this agar such as V. cholerae while sucrose non-fermenters will be blue such as V. parahaemolyticus or V. vulnificus. Also, the presence of black colonies indicates that the microbe can reduce the thiosulfate to hydrogen sulfide (H2S).
Procedure
Inoculate the TCBS agar using the T-streak method with the unknown microorganism.
Incubate at 37oC for 24-48 hours.
6) CARBOHYDRATE FERMENTATION (ACID & GAS PRODUCTION)
Most microorganisms obtain their energy by either respiration (aerobic or anaerobic) or fermentation. Organisms are able to use carbohydrates differently depending on the complement of enzymes the cell has at its disposal. In fermentation, the substrate acts as the electron acceptor such that there is no net change in the oxidation state of the product(s) relative to the starting substrate. In general, fermentation pathways are named for characteristic end products, a property which is widely used in the identification of bacteria. Examples of fermentation pathways are: ethanol fermentation, lactic acid fermentation, mixed acid fermentation, butanol fermentation and butanediol fermentation.
Fermentations are assayed in a tube containing an inverted Durham tube to detect production of gas (CO2). The growth tube contains: a fermentation broth which has all the nutrients required for growth of the microorganism, a specific carbohydrate for fermentation and a pH indictor which changes color at an acidic pH. Fermentation is indicated by the presence of acid (media will turn yellow), in some cases there may also be evolution of gas which will be collected in the Durham tube.
Lack of fermentation does not indicate absence of growth; some microorganism can use other nutrients (such as peptides) as energy sources. These peptides are broken down into amino acids which can be converted to ketoamino acids by deamination which are then metabolized. These types of reactions result in release of ammonia which accumulates as (alkaline) ammonium hydroxide. This type of reaction may also occur if the tubes are incubated too long and all the fermentable sugar is used up. Hence, it is important to read the results in 24-48hr otherwise some of the broths may revert back in color due to the presence of alkaline products.
Procedure
Collect tubes of each broth + sugar (glucose or sucrose or mannitol). Make sure the Durham tube does not contain any air bubbles.
Inoculate each organism in to each sugar.
Incubate at 37oC for 24-48 hours.
Determine the production of acid or acid and gas.
7)NITRATE REDUCTION and DENITRIFICATION
Some organisms are able to use nitrates, instead of oxygen as the terminal electron acceptor, in a process known as anaerobic respiration. Some organisms are able to carry this further by being able to reduce the nitrites, produced as a result of the first set of reactions, further to ammonia, nitrous oxide, molecular nitrogen or some other reduced nitrogenous compounds (hydroxylamine). Denitrification is the process in which nitrate is reduced to molecular nitrogen.
The reaction of nitrate reductase which catalyzes the reduction of nitrate to nitrite can be visualized as:
NO3- + 2H+ + 2e- NO2- + H2O
(nitrate) (nitrite)
other reactions that occur following formation of nitrite are:
NO2- NO N2 (molecular nitrogen)
(nitric oxide)
NH3 N2O
(ammonia) (nitrous oxide)
The reduction of nitrate to nitrite is detected by assaying for the presence of the product (nitrite). Organisms are grown in a nitrate broth medium which contains nutrients and a nitrate source (KNO3) along with a small amount of agar. The agar inhibits diffusion of oxygen, favoring anaerobic growth, therefore utilization of anaerobic respiration. After incubation, reagents A (sulfanilic acid) and B (-naphthylamine) are added to the nitrate broth medium. The presence of a color reaction (red) indicates the presence of nitrite. The color is due to formation of nitrous acid (HNO2), from nitrite, which reacts with sulfanilic acid and -naphthylamine to form a red compound.
Procedure
Inoculate nitrate broths with organisms. Incubate all tubes at 37oC for 24-48 hours.
Examine Durham tube for evidence of gas. Gas may indicate denitrification, alternatively it may be production of CO2 from some fermentative pathway.
Add 3-5 drops of solution A and 3-5 drops of solution B to all nitrate broths (including control). Development of a red color, usually within 30 seconds, indicates the presence of nitrite, and is therefore a positive test for nitrate reduction. No color development indicates that nitrite is not present; absence of nitrite may either be due to nitrates not being reduced (a true negative) or the nitrite formed has been further reduced.
Add a small amount of zinc dust to those tubes that are negative for nitrite. Zinc will reduce nitrate to nitrite. If nitrate remains in the medium it will be reduced to nitrite and a red color will develop. A red color obtained by addition of zinc is a true negative for nitrate reduction. If no red color develops after addition of zinc, then the nitrite has been further reduced to ammonia, molecular nitrogen or other reduced nitrogenous compounds. This is therefore a positive test for nitrate reduction.
Further Characteristics of Gram Negative Bacteria
1) DECARBOXYLASE
Many bacteria produce enzymes capable of decarboxylating specific amino acids, releasing CO2 and alkaline amines. Decarboxylases remove the carboxyl group from specific amino acids, and are involved in the metabolism of several amino acids.
Generally, the reaction is: amino acid (R-COOH) amine(R) + CO2
The decarboxylase you will be determining is:
arginine decarboxylase
L-arginine agmatine + CO2
A control broth of Moeller decarboxylase base medium is inoculated each time as a check of the metabolism of the organism. The decarboxylases are inducible and are only produced in the presence of substrate and at an acidic pH.
the pH indicator in the medium is purple at pH >6.8 and yellow at pH <5.2 Fermentation originally turns the medium yellow due to accumulation of acidic end products, which induces production of the decarboxylases. As decarboxylation of the amino acid occurs the pH of the medium increases on accumulation of alkaline end products resulting in a change in the pH indicator back to purple. Weak reactions appear bluish-gray.
Procedure
In a tube rack collect equal numbers of tubes of Moeller decarboxylase base (contains no amino acid), Moeller decarboxylase base with arginine
Inoculate one of each reaction tube (base and arginine) with each organism. Overlay each tube with about 0.2cm of mineral oil. The mineral oil promotes fermentation with the accumulation of acid end products.
Incubate for 24 hours, continue incubation for up to 4 days.
Record your results (color of medium, and + or - for decarboxylase).
2) UREASE
Urease catalyzes the breakdown of urea by the removal of two ammonia molecules from each urea molecule. This enzyme may play an important role in detoxification of waste products, as well as supplying a source of nitrogen (via ammonia) for amination reactions producing amino acids and nucleotides.
NH2
C=O + H2O 2 NH3 + CO2
NH2
The assay medium for urease activity contains a high concentration of urea and a pH indicator. The ammonia released by the urease reacts in solution to form ammonium carbonate which results in an increase in the pH of the medium.
Procedure
Inoculate the surface of a Urea agar slant with your organisms.
Incubate the slants for 24 hours. Continue incubation for up to 4 days.
A positive result will be seen by a deep-pink to red color in the slant.
3 TRIPLE-SUGAR IRON AGAR (TSI)
The TSI agar test is another important test which allows for a differentiation among groups of the Enterobacteriaceae. Differentiation is based on fermentation of carbohydrates and production of hydrogen sulfide. TSI agar contains lactose (1%), sucrose (1%), and glucose (0.1%). Phenol red, an acid-base indicator, is incorporated to detect formation of acidic fermentation products. The medium also contains sodium thiosulfate as a substrate for hydrogen sulfide (H2S) production and ferrous sulfate to detect any H2S produced. H2S production is detected by formation of a black precipitate (FeS) formed by the reaction of H2S with the ferrous sulfate. Gas production can also be determined by the presence of bubbles in the medium after incubation. Large amounts of gas may cause the agar to be broken and pushed upward. TSI is prepared as a slant, allowing for two different reaction conditions in the same medium: increased surface area for an aerobic environment on the slant region and a butt region in which there is a more anaerobic environment.
Reactions that occur in the various areas of the medium are determined by recording the results at the different sections of the tube, i.e. the slant and the butt.
Alkaline slant/alkaline butt: no carbohydrate fermentation. Peptones in the media are used, resulting in alkaline pH due to production of ammonia. If degradation of peptones is occurring aerobically the alkaline reaction will be evidenced in the slant region; if both aerobic and anaerobic degradation occurs then both the slant and the butt will be alkaline. Organisms that give this type of result are known as non-fermenters.
Alkaline slant/acid butt with or without gas production: only glucose fermentation, with production of a small amount of acid from the 0.1% glucose in the medium. Initially enough acid may be produced to give an acid slant and acid butt, however degradation of amino acids within the slant results in formation of ammonia and therefore an alkaline slant. Peptones in the butt are degraded at a much slower rate due to the anaerobic environment and slower growth of the organisms so it is insufficient to counteract the acid formed by glucose fermentation. Organisms giving the alkaline slant/acid butt reaction are non-lactose fermenters.
Acid slant/acid butt with or without gas production: lactose and/or sucrose fermentation has occurred, with production of larger amounts of acid. The amount of acid produced is sufficient to overcome the effect of peptone degradation so both the slant and the butt are acid. The higher concentration of these sugars, compared with glucose, allows for the continued production of acidic fermentation products. Organisms with acid slant/acid butt reaction are lactose or sucrose fermenters.
Hydrogen sulfide production: is detected by the formation of a black precipitate in the medium. As an acidic environment is generally required for formation of H2S by microorganisms, H2S production is generally found in the butt region of the tube, especially in non-lactose fermenting organisms. Because of the requirement for an acidic environment, the butt should be read as acid even if the usual yellow color is obscure by the black precipitate.
Procedure
Inoculate TSI slants using a straight inoculating needle. Pick up a colony with the needle, stab it all the way into the TSI medium. When the needle is removed inoculate the surface of the slant with a back-and-forth motion.
Incubate the tubes, with the lids loosened slightly, at 370C for 18-24 hours.
Record your results. Record the color of the slant and butt, presence/absence of gas and H2S production.
* record results here as slant/butt reaction in terms of acid or alkaline, +H2S, and production of gas e.g. A/Alk, G, H2S would be acid slant, alkaline butt with gas and H2S production.
Deaminases catalyze the removal of amino groups from amino acids resulting in formation of a carboxylic acid able to enter the central metabolic pathway. Phenylalanine, on deamination by phenylalanine deaminase, forms phenylpyruvic acid, which is detected by adding an oxidizing agent (FeCl3). When ferric chloride reacts with phenylpyruvic acid it changes color from yellow to green.
Procedure
Inoculate, heavily, a phenylalanine agar slant with each organism to be tested.
Incubate for 18-24 hours.
Add 4 or 5 drops of 10% ferric chloride directly to the agar surface. Rotate the tube to dislodge bacteria from the agar surface.
Record your result immediately; the green color fades so read immediately to avoid false negatives.
5) OXIDATION/FERMENTATION (O/F) CATABOLISM
For many bacteria carbohydrates are a prime source of energy. The O/F test can be used to differentiate bacteria based on the whether bacteria are capable of catabolizing a sugar by oxidation or fermentation. The O/F test is carried out with pairs of tubes using a medium containing a small amount of agar (semi-solid), the sugar being tested and a pH indicator (bromothymol blue). Once inoculated, one of the tubes in a pair is sealed with mineral oil to prevent diffusion of oxygen into the medium. Following incubation the ability to oxidize or ferment the sugar is ascertained by a pH (color) change. Organisms that only oxidize the sugar will show a color change in the non-sealed tube but not the sealed one; those capable of fermentation will show a color change in the tube sealed with mineral oil.
Procedure
Inoculate two tubes of O/F glucose with each organism. Use an inoculating needle and stab into the medium.
After inoculation overlay one tube of each pair with 10mm of sterile mineral oil
Incubate at 37oC for 24-48 hours
Record your results.
growth must be present in all tubes - lack of growth indicates that something is missing from the medium.
if only the aerobic tube is acidified, organism is capable of oxidation
if both aerobic and anaerobic tubes are acidified, then the organism is able to ferment the sugar present.
if neither tube is acidified then the organism is unable to catabolize the sugar