Part Ca)Up until the 1940's rubber had only been obtained from the latex of rubber trees(Hevea brasiliensis) in plantations situated in tropical areas such as Malaya and Burma. The demand for rubber increased significantly during world war 2 as rubber was needed for tyres of military vehicles and the supply was interrupted by the conflict between the countries. Scientists in Germany and the US developed synthetic polymers that could replace rubber. Even after the war ended the traditional sources of natural rubber could not meet the greatly increased demand, so synthetic rubbers dominated the market.
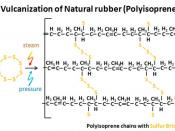
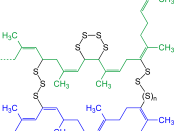
Natural rubber [poly-cis-isoprene(C5H8)n]. the latex consists of a polymer colloid of a monomer called isoprene. The precipitation of the rubber particles from the latex produces an elastic, sticky, gummy mass. In 1839, Charles Goodyear found that by vulcanising (ie. Cross-linking the polymer chains) the natural rubber a non sticky elastic was produced. This vulcanised natural rubber soon became used in a wide variety of products including tyres, insulation, matresses and rainwear.
Rubber production could not keep up with demand, especially from the vehicle industry. A synthetic replacement was needed. And today synthetic polymer production accounts for 80% of the worlds rubber production.
b) Styrene - butadiene rubberStyrene - butadiene rubber (SBR) is the most important synthetic rubber, a copolymer of butadiene (C4 H6) and as with most polymers the raw material is petroleum.
Butadiene rubber BR is important mainly in combination with other rubbers. Compounded with natural rubber and styrene- butadiene in the production of automotive tires. The process for creating styrene-butadiene rubber was through an emulsion procedure that used polymerization as the means of producing a material that had a low reaction viscosity, yet had all the attributes of natural rubber.
The butadiene industry originated in the years leading up to World...