Polypeptides, better known as proteins, carry out nearly all of a cell's functions. A protein is made up of at least one or more amino acid polymers. The necessity of proteins for life is unequivocal, not only do they help to form the structures of cells but they act as enzymes in reactions of the body1.
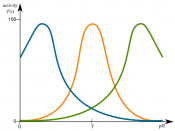
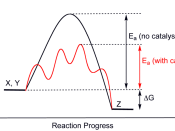
Enzymes are biological catalysts that lower the amount of activation energy needed in carrying out biochemical reactions1. Enzymes are responsible for almost every reaction that occurs in a cell and is named according to the substrate they specifically affect. An enzyme works best under optimal conditions pertaining to temperature, pH level and salt concentration1. In unfavorable conditions enzymes will become denatured and ineffective. In human beings enzymes are most effective in 37 C temperatures, at a pH level of 7.2 (in the stomach a pH of 2 and in the small intestine a pH of 8.5)3
and with a daily intake of five to ten grams of salt4.
Enzymes require either non-protein cofactors such as inorganic substances or organic coenzymes before they can work properly1. Manganese dioxide is an inorganic catalyst capable of breaking down hydrogen peroxide into two H2O molecules and one O2 molecule2. The concentrations of enzymes that are catalyzed during reactions are important, because enzymes can be saturated more substrate will not necessarily affect the speed at which the reactions occur. Once all available enzymes have reacted there is no use for extra substrates1.
Chemical reactions involve the transfer of one or more electrons from one reactant to another1. The process of losing electrons is called oxidization1. The process of gaining electrons is called reduction1. When comparing a protein catalyst to a non-protein catalyst the rate at which the reactions occur will be dependent on the various environmental conditions the substances...