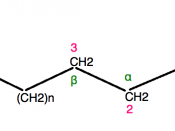
Carbohydrates include sugars and their polymers. Monosaccharides have the general formula of (CH2O)n. The number of these units forming a sugar varies from three to seven with hexases, trioses, and pentoses found most commonly. A sugar can be an aldose or a ketose depending on the location of the carbonyl group. Additional diversity is provided by the spatial arrangement of parts around asymmetric carbons. These small structural differences affect the recognition and interaction of molecules within cells. Glucose is broken down to yield energy in cellular respiration. Disaccharides are joined by glycosidic linkages between two monosaccharides. Maltose, formed with two glucose molecules has a 1-4 glycosidic linkage between the number one carbon of one glucose and the number four carbon of the other glucose. Sucrose has a 1-2 linkage. Polysaccharides are storage or structural macromolecules made from many hundred or few thousand monosaccharides. Starch, a storage molecule in plants, and glycogen, a highly branched polymer of glucose are examples.
Cellulose is the major components of plants cell walls and is the most abundant organic compound on earth. Chitin is a structural polysaccharide formed from modified amino sugar monomers and are fund in exoskeletons.
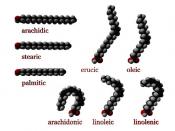
Fats, phospholipids, and steroids are a diverse assemblage or macromolecules that are classed together as lipids based on their hydrophobic behavior. Fats are composed of fatty acids attached to the three-carbon alcohol, glycerol. A fatty acid consists of a long hydrocarbon tail with a carboxyl group at the head end. A triaglcylerol, or fat, consists of three fatty acids, each linked to glycerol by an ester linkage, a bond that forms between a hydroxyl and a carboxyl group. Fatty acids with double bonds in their carbon skeletons are called unsaturated. Saturated fats gave no double bonds in their carbon skeletons and are solid at room temperature.