Many people are attempting to create a solution that will put an end to the environmental problem we know as acid rain. However, is it possible for one to create a solution before they even know the origins of the problem? In the northeastern United States and Canada, acid rain originates primarily from the emissions of carbon dioxide, sulfur dioxide, and nitrogen oxide. These three gases can be converted into acids in the atmosphere.
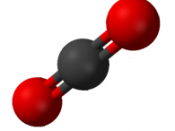
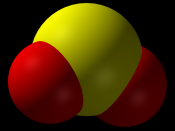
Carbon dioxide is a gas that is released in the largest quantities into the atmosphere. When carbon dioxide (CO2) is released into the atmosphere, it combines with water (H20) in the air to form carbonic acid. Carbonic acid is the weakest of the three acids and causes significantly less damage than that caused by sulfur dioxide and nitrogen oxide. When sulfur dioxide (SO2) is released into the atmosphere, it first combines with oxygen, in a process known as oxidation, and forms a sulfate ion.
This ion then combines with water in the atmosphere and forms sulfuric acid. Sulfuric acid is the strongest of the three acids due to the fact that it is able to completely dissociate in water. Nitrogen oxide (NOx) may also be released into the atmosphere and combine with water forming nitric or nitrous acid. Nitric (nitrous) acid is another strong acid because it is able to almost completely dissociate in water.
Formation of Acids in the Atmosphere Gas Acid Formation Acid Name Carbon dioxide (CO2) CO2 + H20= H2CO3 Carbonic Acid Sulfur dioxide (SO2) 2SO2 + O2 = 2S03 S03 + H20 = H2SO4 Sulfuric Acid Nitrogen oxide (NO2) NO2 + H20 = HNO2 + HNO3 Nitric (nitrous) Acid We now know the three primary gases that form acid rain, however, where do they come from? If we know where these gases...