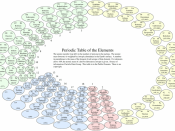
The periodic table of elements is a method of displaying the chemical elements in a table. It was first devised in 1869 by the Russian chemist Dmitri Mendeleev. He intended the table to illustrate periodic trends in the properties of the elements. The layout od the table has been perfected and extended over time because new elements are being discovered and new theoretical models have been developed to explain chemical behavior. Various different layouts are possible, however, the most common forms are still quite similar to Mendeleev's original. The peroidic table provides an extremely useful framework to classify, systematize and compare all the many different forms of chemical behavior. Earlier attempts to list the elements had usually put them in order of atomic mass. Mendeleev's key insight in devising the periodic table was to lay out the elements to illustrate recurring chemical properties and to leave gaps for "missing" elements.
Mendeleev used his table to predict the properties of these "missing elements", and many of them were indeed discovered and fitted the predictions well. With the development of theories of atomic structure it became apparent that Mendeleev had listed the elements in order of increasing atomic number. The main value of the periodic table is the ability to predict the chemical properties of an element based on its position in the table.

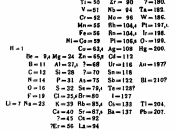



Basic
This essay, although very explanitory is very basic and could use some more detail, perhaps about people who have failed in the past. But this essay is still a nice overview.
0 out of 0 people found this comment useful.