Seperating cyclohexane and toluene by distillation
Aim: Separate two miscible liquids, either by macroscale or microscale process, using simple and fractional distillation. Compare the efficiencies of simple and fractional distillation.
Introduction:
The purpose of this experiment is to learn how to separate two miscible liquids by microscale process. There will be use of simple and fractional distillation. Simple and fractional distillation efficiencies will be compared. The student should have knowledge of how to measure volumes of chemical compounds. In addition, the student should know how to make a bent-tip pasture pipette for microscale distilltions.
Distillation is commonly used in chemistry when compounds need to be seperated because of their differences in their boiling points. Many organic compounds are volatile. Meaning they have high vapor pressures and low boiling points. During distillation, volatile compounds are heated to a boiling in container known as a pot. The vapors produced are colled and reliquefied by passing them through a water cooled condenser.
They are then collected in seperated container called the receiver. This technique is used to remove a volatile solvent from a non-volatile product, seperating volatile product from impurities, or to separate two or more volatile compounds with adequate difference in boiling points
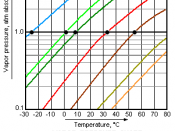
` When liquid is placed in container, some of the molecules evaporate into unoccupied space inside the container. Evaporation continues until there is equilibrium between the molecules leaving and entering the liquid. The pressure exerted by these gaseous molecules on the walls of the container is the equilibrium vapor pressure. The magnitude of this pressure depends on the physical properties of the compound and temperature increases
Liquid is converted to vapor more rapidly by boiling than evaporating. If the heating rate is increased. The temperature of the boiling liquid does not change, but the rate at which vapor...