Sublimation - Chemical Bonding Aim: a) To observe the processes of sublimation and deposition.
b) To better understand chemical bonding.
Introduction: All forms of matter can be changed from one phase to another by adding of removing heat. As most materials are heated they go from a solid phase to a liquid phase and then to a gas. When heat is removed the process is reversed.
Sublimation is an unusual process in which the substance is transformed from the solid to the gaseous phase without passing through the liquid phase.
Deposition is the reverse process.
Materials: Iodine crystals, ice, 100mL beaker, watch glass, candle, ring stand, ring, wire gauze, wooden splints, matches.
Safety Note: Avoid inhaling the iodine vapors.
Work in groups of 4 to minimize the amount of iodine vapor in the air.
Procedure: 1. Your teacher will prepare for you a beaker with a few crystals of iodine.
2. Cover the beaker with a watch glass and place an ice cube on top of the watch glass.
3. Set up the beaker on the ring stand as shown in the diagram.
4. Heat the iodine gently for approximately 5 minutes. Describe what you observe.
The crystals are dissolving and purple gas appears. It rises to the top up to the watch glass.
5. Extinguish the candle.
6. Hold the watch glass by the edge and remove it from the beaker. Do not touch the iodine on the bottom of the watch glass. It will stain your hands. Place the ice cube and water in the sink. Describe what you observe on the underside of the watch glass. The iodine crystallized on the watch glass.
7. Return the beaker and watch glass to the teacherâ≢s table.
Summary Questions 1. Sublimation is the process where a substance changes from a solid to a gas, skipping the liquid phase.
2. Deposition is the opposite process of sublimation, changing from a gas directly to a solid.
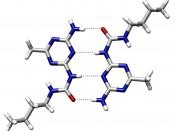
3. Hydrogen bonding causes the difference in behavior between carbon dioxide and water.
Review of Bonding 1. Holds the iodine atoms together in a molecule of I2. Nonpolar covalent bond 2. Holds the many molecules of I2 together in a crystal of iodine. Van der Waalsâ≢ 3. Accounts for the high boiling point of water. Hydrogen Bonds 4. Weak enough to permit CO2 to sublime. Van der Waalsâ≢ 5. Responsible for the extremely high melting point of sand. Network Solid Bonds 6. Permits helium to exist as a liquid. Van der Waalsâ≢ 7. Links H+ with H2O to form H3O+. Coordinate Covalent Bonds 8. Results in substances that are nonconductors in the solid phase and are conductors in the liquid phase. Ionic Bond 9. Links the atoms of a diatomic element. Nonpolar covalent bond 10. Accounts for the ability of metals to conduct heat and electricity.
Metallic Bonds 11. Accounts for the ability of metals to conduct heat and electricity.
Metallic Bonds 12. Holds Mg2+ and Cl- in MgCl2. Ionic Bond 13. Links H+ with NH3 to form NH4+. Coordinate Covalent Bonds 14. Bonds H and F in HF. Hydrogen Bonds 15. Accounts for the high boiling point of HF. Hydrogen Bonds 16. Tetrahedral molecule. CCl4 17. Bent (V shaped) molecule H2O 18. Has the highest degree of ionic bonding. CsCl 19. Has double bonds. CO2 20. This is a dipole H2O