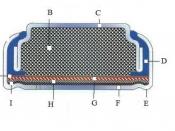
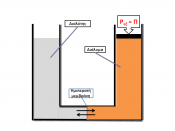
Testable Question Is it possible to observe diffusion across a semi-permeable membrane? Background Research Diffusion is the movement of molecules from an area of higher concentration to that of a lower concentration. Once the particles have reached equilibrium, there is no longer any net movement because a gradient no longer exists. It is the result of the random movement of molecules or Brownian Movement. â¢1 Dialysis is a separation technique used to retain large molecules in one region while permitting smaller molecules to pass through to a different region. In dialysis, the selectivity in movement of molecules is accomplished by placing a semi-permeable membrane between two regions, for example, dialysis tubing. The dialysis tubing is acting as a semi-permeable membrane. Osmosis or Plasmolysis is the movement of water through a selectively permeable membrane from a region of higher water concentration to a region of lower water concentration.
Plant cells have a cell wall as well as a plasma membrane. The cell wall is a rigid structure that is used by the cell to create osmotic pressure within the cell. With the loss of this osmotic pressure the supportive structure of the plant is lost and the plant becomes limp and shriveled. By adding particles such as Na and Cl, the osmotic pressure within the cell decreases. The result of this decrease in osmotic pressure is the shrinking of the cell. The movement of water from the inside of the cell to the outside causes the cell membrane to shrink away from the more rigid cell wall, a process called plasmolysis. This is the diffusion of water out of the cell and into a hypertonic NaCl solution. When placed in a hypotonic solution the plant cell will swell. â¢1 Hypothesis By using the following experiments, it is possible to observe diffusion across a semi-permeable membrane.
Results See attached sheets from lab manual Conclusion Consistent with the hypothesis, the experiments used did in fact account for diffusion across a semi-permeable membrane. In the results for Dialysis, by placing the tubing filled with Karo syrup and starch solution in a solution of water and Lugols iodine, diffusion can be observed. This will be clear by the pockets of starch that turn black on the inside of the tubing. The iodine diffused through the tubing. Also, the tubing became rigid showing that water also diffused into the mock cell causing the turgor pressure to increase.
To demonstrate plant cell osmosis/plasmolysis, a leaf from Elodea was used. When placed in contact with a 10% NaCl solution, osmosis/plasmolysis can be observed. The osmotic pressure within the cell decreases. When observed through a microscope, it can be seen that the protoplasts have moved away from the cell wall and plasmolysis has occurred. The salt solution pulls water away from the cell. The process can also be reversed by placing the cell into a hypotonic solution causing water to diffuse (osmos) back into the cell and when observed through a microscope the cell will be returned to its original condition.