*500 BC- 1720: Alchemists used Ancient Art, during the middle Ages, to discover a substance that would change common metals to gold or silver.
*442 BC: Democritus came up with the theory of atoms, and also he stated that the universe is made up of empty space and that all matter is composed of indivisible atoms.
*384 - 322 BC: Aristotle developed techniques for gathering scientific facts. His progress provided the basis for scientific work more than a millennium.
*1777: Lavoisier, a French chemist, who proved the conversation of matter and explained the mysteries of fire.
*1780s: Coulomb formed the Coulomb's Law; the force one timy charged object exerts on a second one is proportional to the amount of charge on one times the amount of charge on the others and inversely proportional to the square of the distance between them. The formula for this is F= Q1*Q2/r"
*1803: John Dalton, an English chemist, who proposed the Atomic Theory that all matter was composed of tiny indivisible particles known as atoms.
These atoms acquire unique characteristics and weight for different elements. He also declared three types of atoms exist: simple, compound, and complex.
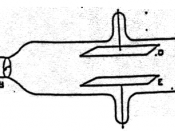
*1870s: Crookes developed the Crookes tube and used it to produce cathode rays for the first time.
*1895: W.K. Roentgen discovered X-Rays.
*1896: Becquerel, a French physicist, found that rays coming from a uraninum ore affected a photographic plate like X-Rays did. The rays were more powerful than those from a sample of pure uranium. His experiment led to the discovery of natural radioactivity.
*1898: The Curies studied and identified radioactive elements.
*1898: J.J. Thomson was the first to measure the electron. He developed the plum-pudding model of the atom.
*1900: Max Planck stated that radiation is quantized.
*1905: Albert Einstein proposed the quantum of light in...