YEAR 11 CHEMISTRY
14
MATERIALS FOR THE
FUTURE - KEVLAR MRS EZARD
TATSUYA SHIIHARA
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
1 | P a g e
Materials of the Future: Kevlar
In this day and age, advances in technology have led to the ability to stare knowingly up at space and
down to the smallest atoms. As a result, many efficient and effective materials have been discovered
and developed, known commonly as
polymers. Polymers are comprised of
monomers, individual molecules, which are
then attached in sequences of chains that
create the polymer. (Encyclopedia Britannica,
2014) With physical properties much better
than other materials, such as higher melting
points, strength and malleability, polymers
can either be naturally occurring (Starch,
cellulose, etc.) or synthetic (Teflon, Kevlar,
etc.). Now applied to nearly all aspects of
everyday life, they can exist in simple
objects such as gloves cards and piping, and also in much more sophisticated apparatuses such as
bulletproof vests.
Chemically known as poly-para-phenylene terephthalamide, Kevlar, one of the foremost polymers
used in the world today, is a cutting-edge, highly effective polymer that has been utilised since the
early 1970's, and is particularly renowned for its
ability to withstand bullet impacts. (Chemistry
in the Military, 2012) First conceived in 1965 by
Stephanie Kwolek, this synthetic polymer uses
much of its intermolecular structure for its
exceptional physical properties, and is
particularly used in the personal protection field,
with Kevlar being the foremost of materials in
bullet-proof vests. ( Marlow Ropes, 2013)
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
2 | P a g e
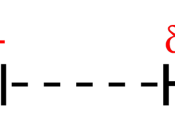
Intermolecular forces are the forces that are between combined molecules that hold everything
together, and there are three types of these forces. These are; London dispersion forces, dipole -
dipole forces and hydrogen bonding. London dispersion forces, al so referred as Van Der Waal's
forces, are weak, instantaneous weak attraction forces that occur between the slightly positive area
of a polar molecule and the slightly negative area of another molecule. This occurs between all
molecules. Dipole-dipole forces exist between two polar molecules as a magnetic force, due to the
various electronegativity's of molecules. (See Appendix A for further information on
electronegativity) They are stronger than dispersion forces, yet weaker than hydrogen bonds. The
strongest bonding of the three, Hydrogen bonding is the result of attraction between a hydrogen
atom and a specific, highly electronegative particle and element. (Virtual Science Fair, 2008)
As can be seen in figure 1, the fabrication process of Kevlar is a condensation reaction, where
monomers join together with a small by-product. The 2 monomers in this case are 1, 4-phenyl-
diamine (para-phenylenediamine) and terephthaloyl chloride, with the by-product being
Hydrochloric Acid. (Chemistry in the Military, 2012) The 2 monomers are held together by dipole-
dipole forces. In order to make Kevlar, the substance that was produced by the condensation
reaction (called the intermediate) must be drawn. (Virtual Science Fair, 2008) This is simply
stretching the product at a certain temperature to strengthen the final product. As drawing the
Kevlar at specific temperatures affects the strength of the final material, it can be inferred that
different heat levels alter resulting Kevlar filaments. Stretching the mould at 400oC, Kevlar-29, the
most common Kevlar used today can be created, whereas in contrast the strongest strand, Kevlar-49,
needs a significantly greater temperature. (Du Pont, 2012) After the Kevlar is drawn, it is spun by
being forced through a spinneret to produce the strands. It is then washed, neutralised and dried
before being wound into coils that are easier for buyers to use, rather than sheets of the material.
(Du Pont, 2012)
Figure 1: Reaction of 1,4-phenylene-diamine (para-phenylenediamine) with
terephthaloyl chloride resulting in Kevlar with a by-product of Hydrochloric Acid.
(Chemistry in the Military, 2012)
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
3 | P a g e
Undoubtedly, Kevlar's superior quality comes from its molecular structure and bonding. Crystalline
properties occur as an inevitable result of the creation of Kevlar, ensuring that the molecules are
straight and ordered. However, a polymer that is crystalline tends to be very strong but does not
have a high malleability factor. (Virtual Science Fair, 2008) Kevlar tends to balance both the factors,
hence resulting in a manipulable material that also has maximised strength. Kevlar has a trans-
conformation ability through its benzene rings and the way each monomer is bonded to the others.
( Putting the pieces together - Microworlds, 2010) As can be seen in Figure 2, this does not allow cis-
conformation, or bends in the molecules, and results in a fully drawn-out fibre as shown in Figure 3,
allowing the polymer to withhold its outstanding level of strength. Hence, Kevlar has an uncommon
property of high tensile strength-to-weight ratio), meaning that it is in reality 5 times stronger than
steel on an equal weight basis. (Du Pont, 2012) (See Appendix A on "tensile strength-to-weight")
Figure 4: Intermolecular forces in Kevlar's
structure (Virtual Science Fair, 2008)
London Dispersion Forces
continuously occurring
Hydrogen bonding
between Kevlar
filaments
Dipole-dipole bonding between
Oxygen, Nitrogen and Carbon
Benzene Rings (See Appendix A for
further information)
Figure 2: Why cis-conformation doesn't form in Kevlar.
(Virtual Science Fair, 2008) Figure 3: Trans-conformation in Kevlar.
(Virtual Science Fair, 2008)
Cis-conformation cannot
occur as the structure is too
tight, with no space for all the
atoms.
A trans-conformation pattern
allows space for all atoms,
with everything stretched out,
and is the form Kevlar takes.
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
4 | P a g e
As can be seen in Figure 4, whilst dispersion forces are helping to constantly hold the fibres together,
although they are very weak and can be easily broken, a huge amount of energy comes from the
hydrogen bonding that holds the strands of Kevlar together. ( Marlow Ropes, 2013) This undeniably
is one of the main reasons that Kevlar has exceptional strength. As a result, all of these
intermolecular properties give Kevlar countless benefits over other polymers. Kevlar can retain the
same amount of strength until 300oC, before starting to decompose at an exceptionally high 500oC.
On the contrary, it can also retain its strength at up to 196oC, with no indication of embrittlement. As
well as this, other than a few powerful acids, virtually all solvents are unsuccessful in degrading
Kevlar. (Virtual Science Fair, 2008)
When asked about the uses of Kevlar, most people tend to think of bulletproof vests and body
armour, yet unbeknownst to most, they are also commonly found as reinforcement in tires and
wheels, as boat hulls, tennis racquet frames, protective casings for fuel cells in aerospace industries,
extra strengthening for fibre-optic cables, various ropes and cables, hoses, helmets and protective
gear for many situations to name a few. (Chemistry in the Military, 2012) (Hamsn Corporation, 2011)
( Marlow Ropes, 2013) It is obvious that all of these applications come accordingly because of the
many intermolecular properties Kevlar exhibits, including the bonding forces, bonding pattern,
trans-conforming properties and balance of strength, crystallinity and flexibility.
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
5 | P a g e
Comparison with another polymer: Zylon
Another polymer that also has a wide range of applications as Kevlar does, is Zylon. Used in areas
such as personal protection, high-altitude balloons, motorsports and yacht rigging, Zylon has a
structure that comprises of dipole-dipole forces that hold it together, as well as constant London
dispersion forces. (Technical F1 Dictionary, 2012) This can be seen in Figure 5.
The study below compares the energy absorption rate of Heavy Zylon, Light Zylon and Kevlar. As can
be seen in Graph 1, on one axis, there is the amount of energy absorbed in Joules and on the
other, the number of layers of the material there are.
Graph 1: Comparison of Kevlar and Heavy and Light Zylon's energy absorption rates
Dispersion forces constantly occurring
Dipole-dipole bonds hold the molecules
together
Benzene Rings
Figure 5: Intermolecular structure of Zylon
(Technical F1 Dictionary, 2012)
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
6 | P a g e
Light Zylon and Heavy Zylon do not have different densities, but amount of denier, which indicates
the fineness of the material. Heavy Zylon is , whereas Light Zylon is only .
When compared, it can be seen from Graph 1 that Kevlar had the lowest absorption rate, with Light
Zylon being slightly higher, and Heavy Zylon clearly outstripping both. On average, Heavy Zylon's
absorbant rate was , whereas Light Zylon's is and Kevlar's was a
mere , as can be deduced from Graph 1.
From Graph 1, it can be seen that Heavy Zylon can absorb up to 3 times more energy than Kevlar
when they both have the same number of layers, making it a much better material in terms of
effectiveness in energy absorption terms. However, it has been revealed that with Zylon, both heavy
and light types tend to degrade rapidly during usage, which makes it very impractical for bulletproof
vests. (Pereira & Revilock, 2008) (Chemistry in the Military, 2012) It can be inferred that this
property is due to the absence of hydrogen bonds holding the polymer together. Only having dipole-
dipole and dispersion forces, Zylon's components obviously can be separated much easier then
Kevlar's as there is less energy holding them together. This is seen in the fact that Zylon is known to
degrade in seawater, ultraviolet and visible light rays and friction. (Pereira & Revilock, 2008) Kevlar is
also known to degrade in UV, however this wall is overcome by the usage of a cover. (Chemistry in
the Military, 2012)
Unlike Kevlar, Zylon is produced by carbon nanotubes, being spun into a fibre. (Technical F1
Dictionary, 2012) (See Appendix A for further information on Carbon Nanotubes) Sacrificing the
malleability of the polymer, this process ensures high strength and therefore a high energy
absorption rate, but also forces the material to degrade at a much faster rate than other polymers.
Hence, Zylon's exceptional strength does majorly not come from the intermolecular forces, but
mostly from as a result of how it was created. (Technical F1 Dictionary, 2012)
With the capability of being reused without any major effects on its energy absorption, Kevlar is
indisputably the better-quality material, and this is obviously why it is currently the most utilised
polymer in the personal-protection field, as well as protective environments such as apparel, covers
and machinery. ( Putting the pieces together - Microworlds, 2010) Its chemical configuration and the
intermolecular bonding that hold it together help to produce a high-strength material that can be
used effectively and efficiently.
The use of Kevlar has most definitely not only transformed our way of thinking by showing us a
hopeful future with nanotechnology, but also has helped our various systems to be more efficient
and effective throughout the world. Without the introduction of Kevlar in the body-armour field, it is
almost certain that many lives would have been lost to this point. The necessary and advantageous
properties that Kevlar possesses for today and the future, both intermolecular and physical, make
Kevlar a highly desirable polymer, and hence it is encouraged to be adopted by industries and
societies worldwide that haven't already taken advantage of this magnificent polymer.
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
7 | P a g e
Appendix A
Term Description
Electronegativity On the periodic table, Fluorine has the strongest electronegativity, and generally, the further away from there, the less electronegativity present. (Dr Nicole Ezard)
Tensile weight-to-strength ratio The maximum stress a material can endure before failure in tension. (Carbon Fiber Tube Shop, 2013)
Benzene Rings A hexagonal arrangement (ring) that consists of 6 Carbon atoms with alternating single and double bonds between them. Each Carbon atom is bonded to a hydrogen atom. (The Free Dictionary , 2010)
Carbon Nanotubes Composed of Carbon atoms linked in hexagonal shapes, each Carbon is covalently linked to three other carbon atoms. (UnderstandingNano.com, 2014)
[TATSUYA SHIIHARA] Term 2 Semester 1 2014
8 | P a g e
Works Cited Virtual Science Fair. (2008). Retrieved May 31st, 2014, from Kevlar:
http://www.odec.ca/projects/2004/clar4c0/public_html/en/fabrication.html
Putting the pieces together - Microworlds. (2010). Retrieved May 24th, 2014, from What Makes
Kevlar So Strong?: http://www2.lbl.gov/MicroWorlds/Kevlar/KevlarPutting.html
The Free Dictionary . (2010). Retrieved June 1st, 2014, from Benzene Rings:
http://www.thefreedictionary.com/benzene+ring
Hamsn Corporation. (2011). Retrieved May 18th, 2014, from Kevlar - High Performance Fibre:
http://www.hamsn.com/blog/kevlar-high-performance-fibre/
Chemistry in the Military. (2012). Retrieved May 22nd, 2014, from Kevlar:
http://advchemorange1.blogspot.com.au/2012/11/kevlar-by-max.html
Du Pont. (2012). Retrieved May 28th, 2014, from KEVLAR® BRAND:
http://www.dupont.com/products-and-services/fabrics-fibers-
nonwovens/fibers/brands/kevlar.view-all.usesapplication.html
Technical F1 Dictionary. (2012). Retrieved May 31st, 2014, from Zylon: http://www.formula1-
dictionary.net/zylon.html
Marlow Ropes. (2013). Retrieved May 26th, 2014, from Physical Properties:
http://www.marlowropes.com/technical/physical-properties.html
Carbon Fiber Tube Shop. (2013). Retrieved May 29th, 2014, from Tube Properties:
http://www.carbonfibertubeshop.com/tube%20properties.html
Encyclopedia Britannica. (2014). Retrieved May 23rd, 2014, from Kevlar:
http://www.britannica.com/EBchecked/topic/315780/Kevlar
UnderstandingNano.com. (2014). Retrieved May 28th, 2014, from What are Carbon Nanotubes?:
http://www.understandingnano.com/what-are-carbon-nanotubes.html
Johns, D., & Guess, J. (2006). Dipole-Dipole Interactions. Retrieved May 21st, 2014, from UC Davis
Chem Wiki:
http://chemwiki.ucdavis.edu/Physical_Chemistry/Quantum_Mechanics/Atomic_Theory/Inte
rmolecular_Forces/Dipole-Dipole_Interactions
Pereira, J. M., & Revilock, D. M. (2008). Ballistic Impact Response of Kevlar 49 and Zylon under
Conditions Representing Jet Engine Fan Containment.
Tatsuya Shiihara NEZ Materials of the Future Term 2 Semester 1 2014
Logbook
Logbook
Date Log
28/04/14 Decided on the use of Sodium Polyacrylate (main) and Cellulose (secondary) for the purpose of this assignment.
09/05/14 Intermolecular forces and electronegativity were found out online (Hydrogen, dipole- dipole, London Dispersion forces), in the Year 11 University Chemistry class (Mrs Natalie Hermon) and the Year 11 Chemistry Class (Dr Ezard)
11/05/14 Further research into Sodium Polyacrylate began.
12/05/14 Research on Sodium Polyacrylate continued.
13/05/14 Research on Sodium Polyacrylate continued.
15/05/14 Unfortunately, due to lack of information, Sodium Polyacrylate was discontinued as the main polymer and Kevlar and Zylon were discovered and decided upon.
16/05/14 Research on Kevlar began. - An introduction was written. - Uses of Kevlar were found. - Information regarding intermolecular forces was found and used.
21/05/14 The monomers that make up Kevlar were found. These were: 1, 4- phenyl-diamine (para-phenylenediamine) and terephthaloyl chloride. Condensation reaction also found.
23/05/14 Properties of Kevlar were found and used in the assignment.
26/05/14 Draft was handed in.
30/05/14 Feedback received.
31/05/14 Diagrams were found for both Kevlar and Zylon. More information about Zylon and Kevlar was found and utilised.
02/06/14 Research was completed for Zylon and Kevlar.
03/06/14 Assignment was concluded.
04/06/14 Assignment was submitted.
- 1. Title Page
- Assignment - Materials of the Future FINAL
- Logbook