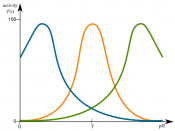
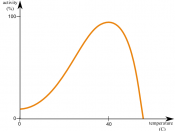
Aim: To see how effectively the enzyme (pepsin) will work in different concentrates of acid. Also to determine if the enzyme becomes denatured.
Prediction: Enzymes are proteins. The functions of an enzyme is determined by the shape of the protein. The arrangement of molecules on the enzyme produce an area known as the active site in which the specific substrate(s) will "fit". It recognises, confines and orients the substrates in a particular direction.
Enzymes allow many chemical reactions to occur within the homeostasis constraints of a living system. Enzymes function as an organic catalysts. A catalyst is chemical involved in, but not changed by, a chemical reaction. Many enzymes function by lowering the activation energy in reactions. By bringing the reactants closer together, chemical bonds may be weaken and reactions will proceed faster than without a catalysts.
Enzymes can act rapidly, as in the case of carbonic anhydrase, which causes the chemicals to react 107 times faster than without the enzymes present.
Carbonic anhydrase speeds up the transfer of carbon dioxide from cells to the blood. There are over 2000 known enzymes, each of which are involved with one specific chemical reaction. Enzymes are substrate specific. The enzyme peptidase (which breaks peptide bonds in proteins) will not work on starch (which is broke down by human-produced amylase in the mouth).
Increase in temperature will speed up the rate of non-enzyme mediated reactions, and so temperature increase speeds up the enzyme mediated reaction, but only to a certain point. When heated too much, enzymes become denatured, because they are proteins dependant on the shape. When/if the temperature drops, the enzyme regains its original shape. Concentration of substrate and product also controls the rate of the reaction, providing biofeedback mechanism.
Changes in pH will also denature the enzyme, by changing the shape...