Letort Vanessa
Student number: 2029474
HEV 4031
Kershaw Paul
ALKALINITY OF AQUEOUS SYSTEMS
INVESTIGATION OF THE CARBONATE /BICARBONATE SYSTEM
AIMS
This laboratory session should enable the operator:
1. To define and understand the importance of alkalinity of aqueous systems.
2. To determine what constitutes a buffer system.
3. To understand the buffering capacity of chemical species such as carbonate and bicarbonate.
4. To gain knowledge of the carbonate/bicarbonate system and its environmental importance in water bodies.
5. To determine carbonate and bicarbonate equivalence points.
6. To practice two different titrations, pH and colour indicators titrations.
7. To calculate the concentration of carbonate and bicarbonate in an aqueous system.
8. To compare accuracy of both titrations.
9. To investigate, identify, and assess error limits associated with those procedures.
INTRODUCTION
Carbon is present in natural waters as the carbonate ion, the bicarbonate ion, and as dissolved carbon dioxide.
Carbonate ions come from the weathering of carbonate rocks, leaching in surface waters.
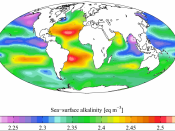
Carbon dioxide is present as a gas in the atmosphere. Carbon dioxide is a product of respiration of plants and animals and is released into our atmosphere when carbon-containing fossil fuels are burned in air. It makes up for only 0.03% of the total mass of the atmosphere. This fraction seems small but the function of CO2 in global biological and environmental systems is of prime importance (Loewenthal & Marais, 1978). Despite its low atmospheric concentration, carbon dioxide is normally abundant in natural waters because its solubility is more than 30 times higher than that of oxygen. The marine carbonate system represents the largest carbon pool in the atmosphere, biosphere, and ocean and is therefore of primary importance for the partition of excess carbon dioxide produced by man.
Carbon dioxide in the water dissolves, making carbonic acid, which lowers the pH.