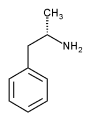
Amphetamines are part of a class of drugs called stimulants that affect the central nervous system. It is also known as 1-methyl-2-phenethylamine, 1-phenyl-2-aminopropane, and Benzedrine. It is made up of a 50/50 mixture of the l and d isomers. The l isomer affects the body, and the d isomer affects the brain. It boils at 200úC and is soluble in alcohol and ether, slightly soluble in water. It was first created in Germany in 1887. In the 1930s and 40s amphetamine was used in the treatment of asthma, depression, obesity, and narcolepsy. Amphetamine's chemical structure is very similar to the neurotransmitter Adrenaline.
Chemical structure of Amphetamine,
C H CH CH(NH ) CH
Method of Preparation
There are many ways of creating amphetamines. One way is to take two mols, for example, 270 grams, of racemic amphetamine base to react with one mol (150 grams) of d-tartaric acid, forming a dl-amphetamine d-tartrate, or a neutral salt.
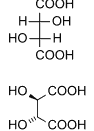
The neutral salt obtained is dissolved by the addition of about 1 litre, of ethanol, and heating to boiling point. The solution then must be cooled to room temperature with occasional stirring to effect crystallisation. The crystals are filtered out and will be found to contain laevo enantiomorph, which is the left of a pair of crystals that are a mirror image of each other. It is then recrystallised and the predominance of the l-enantiomorph is found to be increased and the process is repeated until no further change is effected and a concentration of 8 grams per 100 cc of aqueous solution is obtained. Finally the product obtained is l-amphetamine d-tartrate. d-amphetamine may be recovered from this by the addition of tartaric acid for the formation of acid tartrates and separation of d-amphetamine d-bitartrate by crystallisation.
Properties and Chemical Reactions of...