Artem Ilyayev
Ap Bio
1) An element is to a(n) ___________ as an organ is to a(n) _______________
Answer - b - compound; organism
2) In the term trace element, the modifier trace means
Answer - a - the element is required in very small amounts.
3) Compared to 31P, the radioactive isotope 32P has
Answer - b - one more neutron
4) Atoms can be represented by simply listing the number of protons, neutrons, and electrons - for example, 2P+; 2n0; 2e- for helium. Which atom represents the 18O isotope of oxygen?
Answer - b - 8p+; 10n0; 8e-
5) The atomic umber of sulfur is 16. Sulfur combines with hydrogen sulfide. Based on the electron configuration of sulfur, we can predict that the molecular formula of the compound will be
Answer - c - H2S.
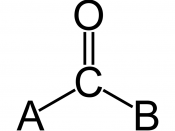
6) Review the valences of carbon, oxygen, hydrogen, and nitrogen and then determine which of the following molecules is most likely to exist.
Answer - b -
7) The reactivity of an atom arises from
Answer - b - the existence of unpaired electrons in the valence shell.
8) Which of these statements is true of all anionic atoms?
Answer - a - the atom has more electrons than protons.
9) What coefficients must be placed in the blanks so that all atoms are accounted for in the products?
C6H12O6 = __ C2H6O + __CO2
Answer - b - 2;2
10) Which of the following statements correctly describes any chemical reaction that has equilibrium?
Answer - b - the rate of the forward reaction equals the rate of the reverse reaction.
CHAPTER 3
1) What is the best explanation of the phrase "fitness of the environment", as use I this chapter?
Answer - d - Life as we know it depends on certain...