Enzymes are catalysts. Catalysts are substances that speed up chemical reactions. The rate at which the reactions increase are influenced by their environment. Temperature and pH are two important environmental factors.
The relationship between structure and function of an enzyme is also important. The specificity of an enzyme comes from it's structure, or shape. A compatible fit between the shape of the enzymes' substrate and active site attributes to its specificity. The functions of an enzyme will vary because of the different structures. For example one function of a specific enzyme is to convert substrate to product. The rate at which the substrate is converted depends on the concentration level of the substrate. The functions of enzymes vary because of the enzyme's structure.
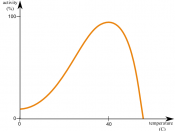
Increasing temperature, up to a point, increases the speed of an enzymatic reaction. This is partly because substrates collide more frequently when the molecules move quickly.
Once the optimal temperature for an enzyme is reached the speed of the enzyme activity drops sharply. The enzymes then begin to disrupt the ionic, covalent, and other weak interactions that stabilize the active conformation. This causes the enzymes to denature. The temperature that allows the most molecular collisions without denaturing the enzyme is the optimal temperature for which reactions occur the fastest.
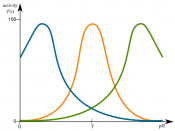
Each enzyme also has an optimal pH level at which the enzymes are most active. As pH raises so does the enzyme activity, until it reaches that optimal level. For most enzymes this is usually between a pH of 6 and 8. However there are some exceptions. After the optimal level, the enzyme activity decreases rapidly.
A simple experiment could be set up to produce the same results as seen in the pH graph. The hypothesis should be similar to the one as follows: If pH raises...