Letort Vanessa
Student number: 2029474
HEV 4031
Kershaw Paul
DETERMINATION OF AVAILABLE PHOSPHATE IN WATER
SPECTROPHOTOMETRIC PROCEDURE
AIMS
This laboratory session should enable the operator:
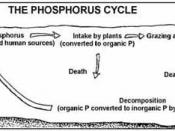
1. To gain knowledge of the phosphorus cycle.
2. To understand the importance of the phosphorus element in biological systems and environmental systems.
3. To identify the origins of phosphorus pollution and its impact on water ecosystems.
4. To understand the process of eutrophication of water, and the role played by phosphorus.
5. To gain experience in the practical application of colorimetry as an analytical technique.
6. To use the spectrophotometer method to determine the concentration of phosphate in different natural waters and an unknown sample.
7. To introduce the method of preparing standards by serial dilutions and to use these standards in the production of a calibration curve.
8. To analyse and identify the errors limits inherent of the procedure.
9. To compare results with guidelines on water quality.
INTRODUCTION
Phosphorus is a non-metallic element, belonging to group V of the periodic table. There are three allotropic forms of the phosphorus element, red, white, and black. All phosphorus allotropes are very reactive (Concise Science dictionary, 1984).
Phosphorus is of primary importance in biological systems. Phosphorus is the key to energy in living organisms; it is phosphorus that moves energy from ATP to another molecule. Adenosine triphosphate is important in the storage and use of energy and a key stage in the Kreb's Cycle. Therefore, the availability of phosphorus is a key factor controlling photosynthesis, thus a key factor at the base of any food chains. Phosphorus is also contained in DNA and RNA, binding desoxyribose sugars together in DNA, and binding ribose sugars together in RNA (Porteous, 2000).
Furthermore, a low level of available phosphorus is a major limiting factor for plants growth,