AbstractThis experiment investigated the amount of DNA extracted from strawberries. This was done by using the independent variable of alcohol to affect the dependant variable of the amount of DNA extracted. This was done to find out if primary or secondary alcohol would create more DNA precipitate than the other. For this the primary alcohols used were; methanol and ethanol, and the secondary alcohol was; isopropyl. Of this the secondary alcohol, isopropyl was discovered to be the most effect alcohol to make DNA precipitate, as it produced the most amount of DNA. This investigation of extracting DNA is significant due to the study taken providing understanding and knowledge on DNA; this allows people to find out information of the cell structure, and what DNA does.
IntroductionThe aim of this experiment is to find the effects different types of alcohol, primary or secondary, has on the quantity of DNA extracted from strawberries.
The results should show that methanol and ethanol would have similar results due to both being primary alcohols. Isopropyl would have better result due to it being a secondary alcohol.
DNA or deoxyribonucleic acid is the nucleic acid molecule that stores the hereditary information of the organism and is indirectly responsible for cellular structure and metabolism (Aubusson, Kennedy, Snyder 1990: 474). The main role of DNA acid is the long term storage of information. DNA is a set of blueprints or a code since it contains the instructions needed to construct other components of cells, such as proteins. The DNA divisions that carry this genetic information are called genes; other DNA sequences have different structural purposes, for example to create a body part, like an arm or a leg. Because of this, DNA provides information on hair colour, eye colour, skin colour, etc...
DNA extraction is important because it enables people to see the information about DNA and shows how complex information is stored. DNA is found in chromosomes inside the nucleus. This DNA is wrapped around a ball shaped histone protein. The DNA molecule is a double helix structure and is very long; this is the building block for life.
It is important to achieve maximum DNA out of the results as the increased amount of DNA that is collected, the better it is able to be studied in some depth.
The reasoning behind the investigation was to study the amount of DNA extracted when using different types of alcohols. This information is needed, to know the best possible way of obtaining maximum amounts of DNA from the cells to optimise the protocol. The different types of alcohol were methanol and ethanol (primary alcohols) and isopropyl (secondary alcohol).
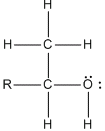
The alcohol allows for DNA's fragments to precipitate/stick together this produces a blob of DNA which can be spooled out and examined. There are different types of alcohols; primary, secondary and tertiary. The construction of primary, secondary and tertiary alcohols depends on the different amounts of carbon attached to the alkyl groups. Some examples of a primary alcohol are; ethanol and methanol, secondary alcohol; isopropyl, and tertiary alcohol; tert-butyl. DNA is not soluble in alcohol. So when alcohol is added to the mixture, mixture, except for DNA, stay in solution while the DNA precipitates out into the alcohol layer. Also alcohol is less dense than water, so the alcohol stays on top of the mixture without mixing in.
The importance of finding the most effective way of obtaining the most amount of DNA from different primary and secondary alcohols is so that it can be compared. The detergent will act and dissolve or separate the lipid components of the cell membrane; this gives access to the proteins and nucleic acids within the cell. The detergent cells are similar to phospholipids. The similar district of both detergent and phospholipids work together, disrupting the structure of the cell membrane and causing it to break apart. The salt helps to break down the cell membrane by denaturing the proteins in the membrane. As salt, (NaCl) detaches in an aqueous solution to form Na+ and Cl-, initially this helps to break down the cell wall and nuclei. The reason why the DNA is heated to 65ðC as it speeds up the precipitate, also denature the DNA enzymes that cause shearing of the DNA, and like salt, heat is also used to disrupt the cells. The enzymes in the meat tenderiser are called papain, it is used to break up the histone proteins, into smaller pieces this detaches the DNA, allowing the DNA to uncoil and be seen.
Material/methodBefore starting the experiment, ensure that the methanol, ethanol and isopropyl are in the freezer (at -10ðC). Also set up the filtering apparatus (see figure 1). Next blend 125g of Strawberries; Measure our 100mLs of the strawberry mixture and mix in 100mLs of distilled water, then put the mixture into the filtering apparatus, and leave to filter place filtrate mixture into a beaker. Fill a large beaker with hot water and place the smaller beaker inside (see figure 2) until the mixture is heated to 65ðC. Take out the mixture and stir in 1gram of salt, 1g of meat tenderiser and 1mL of detergent. Pour 5mL of the now filtered and heated mixture into each of the 3 test tubes. In test tube 1add 5mL of methanol gently and slowly down the inside of the test tube. In test tube 2 add 5mL of ethanol. And in test tube 3 add 5mL of isopropyl. Allow for the DNA to precipitate and observe what is occurring. Once the DNA has precipitated it will look like cottony fluff in the alcohol or mixture. Collect the DNA using a wooden cocktail skewer, and place in labelled and sealed containers with 1mL of water and store in a fridge (at 4ðC). Record each amount extracted. Repeat the method three times then run through a 1% agarose 1x TAE gel. All results observed should be recorded in a table.
ResultsAlcohol QuantityObservationsMethanol+This bubbled and formed really quickly. It formed on top of the strawberry mixture. There was small amount of DNA, and wasn't too thin. (See figure 3) Gel Electrophoresis failed.
Ethanol+There was small amounts of DNA and was not as clumped together in the mixture, and it was thin. It formed in the middle of the alcohol. It forms slower than the others. (See figure 4) Gel Electrophoresis failed.
Isopropyl+++There was a large amount of DNA extracted and it was collected towards the bottom half of the alcohol part. It formes at a steady pace, and was quite thick. (See Figure 5) Gel Electrophoresis failed.
Key:-+ Small amount++ Middle amount+++ Large amountDiscussionThe results show that the hypothesis is confirmed; methanol and ethanol would have similar results due to both being primary alcohols. Isopropyl would have a better result due to it being a secondary alcohol. With this the aim has also been achieved, as methanol and ethanol, produced minimal results compared to isopropyl.
In the methanol, overall from the three repeated tests it produced a bit of DNA, which was thick and the strands were quite long. It was also formed very quickly around the air bubbles trapped in the alcohol and strawberry purée. This formed on top of the strawberry purée.
With the ethanol, overall from all the tests the DNA produced was little, and very thin, which made it difficult to retrieve. This precipitated quite slowly and formed more in the middle of the alcohol.
The isopropyl, from the results recorded from all three tests showed excellent results, with lots of DNA precipitate and the DNA was thick and in very long strands, that were easy to collect. This formed towards the bottom half of the alcohol, and at a steady pace.
Even though ethanol and methanol are both primary alcohols, some of the results were similar; however methanol had a thicker DNA precipitate to ethanol. This is due to most primary alcohols have a carbon which carries the -OH and is only attached to one alkyl group. Ethanol: CHó-CH2-OH. And that is an exception to methanol. It is still counted as a primary alcohol even though methanol (CHó-OH) has no alkyl groups attached to the carbon with the -OH. While isopropyl, a tertiary alcohol is made up of the carbon with -OH group attached directly to two alkyl groups, while the primary alcohols are only attached to one.
When the DNA was taken out of the Gel Electrophoresis, it did not show any signs to measure the amount of DNA. This means that the gel electrophoresis failed. The gel is meant to be used to compare roughly how much DNA is present. After loading the DNA into the gel it would be let run for about an hour, after which it would usually be stained with ethidium bromide, which binds to DNA. However, the school does not stock this so methylene blue was used instead. After this it is placed on a UV light box and photos can be taken to compare.
There are a few possible reasons for the gel to fail; the gels may have been too thick, the fridge may not have been cool enough, or the methylene blue solution may have been too concentrated. However, to improve future developments with a gel, the following might be considered, gels might have been better off being left in the stain for less time, using a more dilute stain, staining for less time (checking gels every 30min-1hr) or to possibly use thinner gels.
In further tests on DNA extraction, it would be recommended that not only are the gels modified but also some other variables. These could include; adding more distilled water to the strawberry mixture so it isn't as thick and take so long to filter, also on this note, having a thinner filter paper could also be benefited from. For more comparison between the alcohols, more primary and secondary alcohols should be used, as well as possibly have a tertiary alcohol for further comparison. Other modifications could be made by doing each extraction with a different batch of strawberries, as well as leaving more time for the DNA to precipitate.
ConclusionIn conclusion, this experiment achieved the aim and conformed to the hypothesis, by finding the most effective alcohol for extracting DNA. The results and discussion show that the hypothesis has been confirmed, as isopropyl, a secondary alcohol was the best alcohol for extracting DNA. This was because it causes more DNA to precipitate. In the future, the recommendations for further improvement should be followed, to gain better results.
ReferencesPeter Aubusson, Eileen Kennedy, Wade Snyder, (1999), Glossary- DNA (date used- 29/03/09)Jim Clark, (2003) Introducing Alcohols, (date accessed- 23/03/09)Wikipedia, (29 March 2009) DNA, (date accessed- 21/03/09)Access Excellence, (last modified- unknown date) Introduction to a DNA Extraction, (date accessed- 21/03/09)University of UMBI, (2008) An Introduction to the DNA Extraction Lab (date accessed- 21/03/09)ThinkQuest, (2004) DNA- Deoxyribonucleic Acids, (date accessed- 19/03/09)Microbiology, (2008) Why is DNA Important, (date accessed- 19/03/09)