AP Biology Chapter 5
5.1 Macromolecules
Macromolecules are polymers built from polymers
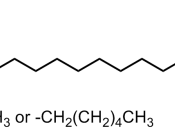
Carbohydrates, Proteins, and nucleic acids are chain like molecules, while lipids are polymers them selves
Polymers are long molecules consisting of many similar identical building blocks linked by covalent bonds = monomers
Polymers
In cells, the breakdown of polymers are facilitated by enzymes, proteins that speed up reactions
Monomers are linked by a reaction in which two molecules are covalently bonded with the loss of a water molecules, also known as a dehydration reaction
When a bond forms between two monomers, each molecule contributes a part of a water molecule, either a hydroxyl (OH) or a hydrogen (H).
Polymers are disabled by hydrolysis(water breakage) , a process in which water is added to the polymer and therefore breaks the bond between the monomers, this process occurs in our digestion
Polymers are made from about 40-50 monomers, and the key to infinite possibilities is arrangement
Carbohydrates
Serve as fuel and building material, include sugars and polymers of sugars
The simplest carbohydrates are the monosaccharides, or simple sugars(monomers)
Disaccharides are double sugars consisting of two monosaccharides joined by covalent bonds
Macromolecules are polymers called polysaccharides
Sugars
Monosaccharides have molecular formulas of the unit CH2O.
Glucose is the most common one (C6H12O6)
Has a carbonyl group and multiple hydroxyl groups
Depending on location of carbonyl group, it's a ketose(middle of carbon skeleton or an aldose (end of carbon skeleton)
Can also classify sugars by the size of the carbon skeleton which ranges from 3-7 carbons
Triose(3 carbon) Glycerldehyde and Dihydroxyacetone
Pentos(5 carbon) Ribose and Ribulose
Glucose, Fructose, Galactose have 6 carbons
Another source of diversity is by their arrangement for example, Glucose and Galactose only differ in the asymmetric carbon.
In solutions, Glucose molecules form rings
Monosaccharides are a major...