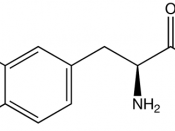
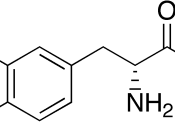
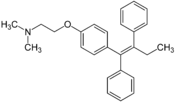
IntroductionProgressive degeneration of nigrostriatal dopaminergic pathway leads to the depletion of striatal dopamine which causes ParkinsonÃÂs disease (PD). This disease is a disorder that affects the nerve cells or neurons in the part of brain that controls the movement. The neurons named dopamine which makes some chemicals in the body die or do not work properly. No one knows about the damages made by these cells. (Parkinson's disease, 2008)The administration of the pharmacological agents L-DOPA could bring back a normal dopaminergic transmission. PD causes due to some motor deficits such as tremor, rigidity and bradykinesia, L-DOPA reduces these motor deficits. The treatment with L-DOPA twice in a day shows the manifest similar motor response change after the chronic treatment by Rodents, which renders hemiparkinsonian following brain administration of 6-hydroxydopamine (6-OHDA).
Some non-human primate studies suggest that the sensitivity of corticostriatal glutamatergic neurotransmission is increased by the motor response complications from the pulsatile nonphysiological stimulation of dopaminergic receptors on the striatal spiny neurons.
The development of response alterations produced by L-DOPA implicates that the activation of calcium/calmodulin dependent protein kinase II, cyclic AMP dependent protein kinase A and tyrosine kinase signaling cascades in association with the enhanced phosphorylation of NMDA receptor subunit in rat striatal neurons.
The development of characteristic response alterations could influence an inhibitor of PKC. The PKC is divided into three groups which include classical or conventional PKCs. Tamoxifen which works as a PKC antagonist is an anticancer and antiestrogen drug. Tamoxifen is a suitable drug for evaluating the therapeutic potential of PKC antagonist in the treatment of L-DOPA. The purpose of this study is to prove that the side effects of L-DOPA could be reduced with the use of tamoxifen.
MethodAn experiment has been done by the researchers to prove the reduction in side effects of L-DOPA. The method and its result are discussed in this section. Some rats weighing 150-200 grams and monkeys weighing 3.5-7.5 kg were kept in a house. Rats were allowed to use water and food but monkeys were fed with biscuit diet twice a day and had a free access of water. All the animals were injected with 6-OHDA to cause ParkinsonÃÂs disease. After three weeks, all the hemiparkinsonian rats were screened by a swing test. The rats were considered further who 100% turns ipsilateral to the lesion. The chronic L-DOPA treated rats were given apomorphine and screened again. Rates who got rating >100 contralaterals turns in 1h, these rats were considered again for testing.
Rats were injected with L-DOPA twice a day (for the study of tamoxifen), for a period of 21 days, after two days of screening with apomorphine. The motor responses were checked at the beginning and at the end of the process of injecting L-DOPA. Rats were selected for further studies who had >15% reduction in response after 21 days when compared to day 1.
The animals were treated with vehicle (tween 80/glycerol, 50/50%, p.o.;n=4)/tamoxifen (5 mg/kg prophylactive treatment; n=5). Vehicle/tamoxifen treated rats were treated with acute L-DOPA. Some of the rats from 6-OHDA lesioned and 6-OHDA/chronic L-DOPA were given acute L-DOPA. The injected rats lost striatal tissue from both the hemisphere. A buffer solution of NaCl, EDTA, EGTA NaF NaPPi, Na3VO4, leupeptine, aporotonin, pepstatin A and dd H2O homogenized striatal tissue.
A western bolt analysis of striatal homogenous occurred with the help of 2ÃÂSDS sample buffer, vortexing, aliquots, 4-12% linear gradient polyacrylamide gels. Protein were separated and transferred to a nitrocellulose membrane. A comparison between the migration levels of the immunoreactive bands and standard marker See-Blue estimates the molecular mass of the proteins.
The effect of tamoxifen could be examined by chronic L-DOPA treatment on motor response alteration on day 1, 21, 22, Tamoxifen was administered orally 1 h prior to accurate L-DOPA administration, on the 22nd day of L-DOPA treatment. The above treatment is called the palliative drug treatment. After apomorphine rotational screening and acute L-DOPA rotational screening, animals are divided in two groups. One group follows the treatment with L-DOPA with vehicle and second group follows the treatment with L-DOPA with tamoxifen. This process is called prophylactic drug treatment.
Monkeys were injected with 0.5-1 mg/kg 1-methyle-4-phenyle-1,2,3,6-tetrahydrophiridin once in a week. The process continues until definite parkinsonian features. All animals were treated with oral L-DOPA/carbidopa. A dyskinetic does of L-DOPA was fixed for every animal for the period of one week. All animals were administered with the dose of 0.03, 1.0 and 3.0 mg/kg of tamoxifen. A dose of L-DOPA with an increased does of tamoxifen was administered. All the monkeys were transferred to a room.
The monkeys were observed until their clinical state got stabilized. Their medical records and neurological interventions were examined by the NIH veterinarians. The assessment of parkinsonian severity was based on ÃÂDisability ScaleÃÂ and assessment of dyskinesias was based on ÃÂMovement ScaleÃÂ.
ResultsThe injected 6-OHDA resulted in dopaminergic neuronal denervation of the left striatum 6-OHDA and 6-OHDA plus chronic L-DOPA as evidenced by the absence of an immunoreactive band for TH in both types of candidates. It was found that there was a slight increase in PKC eta in the left as compared to the right striatum. The nigrostriatal pathway got destructed with the use of 6-OHDA and chronic L-DOPA treatment. Western Blot detected û and PKC elipson protein level in the rat striatum. A reduction was observed in PKC elipson.
The reduction in protein expression of PKC lambda was indicated by rats treated with in chronic L-DOPA plus tamoxifen comparison to acute L-DOPA. There was no significant alteration in PKC lambda on day 21. There was no difference in the PKC immunoreactivity. The acute L-DOPA challenge was received by both vehicle and tamoxifen treated rats. The protein expression was evaluated by the effect of the treatment of chronic L-DOPA with tamoxifen. The PKC immunoreactivity for both vehicle and tamoxifen was same.
When the treatment of L-DOPA with tamoxifen on day 21 and treatment of L-DOPA on day 22 was compared, it was found that the administration of medium dose of tamoxifen ameliorated the response shortening induced by L-DOPA therapy. The motor responses were induced by the effect of tamoxifen. On the 22nd day, tamoxifen group was higher. The effects of tamoxifen group on day 22 and the effects of L-DOPA on day 21 were same.
The vehicle or tamoxifen was given orally 1h prior to L-DOPA for 21 days. When the performance of vehicle on day 14 and 21was compared with day 1 and day 2, the performance was significantly lower. When the performance of tamoxifen on day 14 and day 21 was compared with day 1 and day 2, there was no difference. The animals which were given tamoxifen with L-DOPA had a longer response to L-DOPA in comparison to those animals which were given vehicle with L-DOPA. When tamoxifen is given alone to parkinsonian, it has no effect, but when it is given with L-PODA, it reduces the appearance of dyskinesias.
DiscussionThe experiment shows that how some PKC isoforms are linked with the genesis of L-PODA. Tamoxifen with L-DOPA could reduce the side effects of L-DOPA. Tamoxifen reduced the responses produced by chronic L-DOPA therapy in parkinsonian rats and other non human primates. Tamoxifen was able to prevent the development of responses in 6-OHDA lesioned hemiparkinsonian rats.
The monkeys injected L-DOPA coadministered tamoxifen with L-PODA. The appearance of dyskinesias is reduced by tamoxifen in PD primates. A potential tolerance to affect tolerance should be considered to find some result since tamoxifen was given in an increased dose and the difference between periods was two days. Tamoxifen does not produce any alteration in motor response and has no affect on parkinsonian sign. The only factor which plays a role in pathogenesis of motor responses is central mechanism, which is influenced by tamoxifen.
The phosphorylation of GluR1 subunit of the AMPA receptor was induced by PKC. The motoer dysfunction was induced by the critical role pf glutamatergic corticostriatal sensitization in the genesis of L-DDOPA. Western blot analysis shows the normalization of the protein expression of PKC lambda. Tamoxifen is administered by the effects of normalizing. The same does of tamoxifen which was given with L-DOPA would not generate any response without the involvement of acute L-DOPA. The process shows that PKC isoforms have a profound role in motor response. A similar downstream signaling cascade involving the lambda and elipson isoforms could be responsible for the aberrant cellular activation.
The observation is based on the effects of 6-OHDC on rats and MPTP, which creates parkinsonian sign in animals, before the effect of tamoxifen. Tamoxifen is able to stimulate dopamine release, which is one of its pharmaceutical properties. Chronic L-DOPA therapy is used to increase the expression of preproenkephalin mRNA in normal monkeys.
The study shows a decrease in the PKC epsilon and increase in PKC lambda. The reactivated PKC epsilon and activated PKC lambda contributes to the development of motor responses. The motor responses are developed by the corticostriatal glutamatergic synaptic effects. The expressions of PKC epsilon were induced by L-DOPA only in the left hemisphere. The pathogenesis of PD and dyskinesias involves striatal asymmetry. Tamoxifen has multiple uses, it is also used as an anti-breast cancer drug.
The report supports the view that enhancing L-DOPA wit Tamoxifen could reduce the side effects of a medicine like L-DOPA. Tamoxifen is a potential therapeutic agent which is used to prevent L-DOPA induced dyskinesias in PD patients.
ConclusionThe use of tamoxifen reduces the side effects of L-DOPA. The experiment which was performed on animals such as rats and monkeys, indicate the role of tamoxifen to reduce dyskinesias in PD patients. When L-DOPA is given alone to an infected animal, than it would create some side effects and the rate of treatment without tamoxifen is very slow. When an increasing dose of tamoxifen is given with L-DOPA, it helps in reducing the side effects and induces the rate of recovering of the motor response.
ReferencesParkinson's Disease Reviews: 2002, (2001), Retrieved February 23, 2008, from http://lansbury.bwh.harvard.edu/pd_reviews_2002.htmMedical and surgical treatment of Parkinson's disease, Retrieved February 23, 2008, from http://www.postgradmed.com/issues/1999/08_99/conley2.htmNational Institute of Neurological Disorders and Stroke National Institutes of Health, (November, 2004), Retrieved February 23, 2008, from http://www.ninds.nih.gov/disorders/parkinsons_disease/parkinsons_research.htm#drugParkinson's Disease, (2008), Retrieved February 23, 2008, from, http://www.nlm.nih.gov/medlineplus/parkinsonsdisease.html#cat3