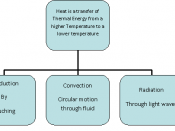
Heat is the amount of energy generated by the internal motion of the molecules of a substance, while heat transfer is the movement of heat from a warmer object to a cooler one. This theory of heat transfer can be proven by observing the cooling of water in a glass. The hot water tries to transfer heat to the cooler glass. Therefore, the two substances try to reach equilibrium. There are three different types of heat transfer: conduction, convection and radiation. The method of transferring heat between molecules is specific to each type of heat transfer. Conduction is the transfer of heat by direct contact of molecules.
Depending on which type of heat transfer is happening, there are very specific ways to suppress the transfer of heat. The problem is, what may be good for one type of transfer is bad for another. In hopes of figuring out the best way to insulate boiling water, we were given an assignment to build a heat saving device.
The key is to figure out what combination of insulation will benefit all three transfers while doing the least amount of 'damage'
The objective of this experiment is to build a device that optimizes the insulation and minimizes the amount of heat lost from 150mL of water originally at 100 degrees Celsius. The water is boiled in a beaker, then poured into another beaker and sealed into the device for 20 minutes. The device that keeps the water the hottest wins the class competition.
The heat transfer I chose to study was conduction. The basic principle of this heat transfer is when two molecules hit against each other, the one traveling faster will give some of its heat energy to the slower moving one, because heat moves from hotter objects to cooler objects.

Excellent work
For a grade 8 essay this is without a doubt very good and you deserve the A+. You have expressed your idea's very well and it is more than obvious that you understand the topic very well. However a word of advice when you do essay's in future (just to pick up more marks) try to stick with third person terms particularly in science based assessments as teachers are very pushy about it ( I saw you refer to second person terms occasionally e.g. you or your ). Furthermore try to narrow down and be as concise as possible this way you can add in more info and not seem like you are blabbing away. But besides that you have done an excellent job. Keep it up and I enjoyed reading this essay its helped me a lot in understanding Thermal conductivity and thermal insulators in a much more simpler way then going through other web sites.
0 out of 0 people found this comment useful.