Aim
To illustrate how gas chromatography (GC) may be used for the identification and quantification of individual constituents of a complex natural product.
Introduction
Gas chromatography (GC) separates mixtures into their individual components and is particularly useful for volatile and semi-volatile compounds. GC is used in the analysis of a wide variety of samples, including paints, polymer additives, solvents, hydrocarbons, pesticides, and drugs. Samples are introduced to the stationary phase of the capillary column as vapours or dissolved in solution. As the column is heated, an inert gas acting as the mobile phase is passed through the column. Each compound in the sample is carried through the column at a different rate, separating the sample into its individual components. Each component is detected as it emerges from the column by a flame ionization detector. The "retention time" spent in the column for each component is used for identification and the intensity of the signal for each component can be used to quantify the compound of interest.
When reference standards can be analyzed under identical conditions, GC profiles can also be used as fingerprints of complex mixtures.
Theory
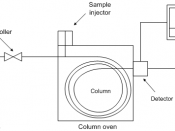
The GC consists of an injection block, a column, and a detector. An inert gas flows through the system. The injection chamber is a heated cavity which serves to volatilize the compounds. The sample is injected by syringe into this chamber through a port. Once inside, the sample becomes vaporized and is carried out of the chamber and onto the column by the carrier gas.
The column is an integral part of the GC system. On the outside, all you see is a long stainless steel tube, which is coiled. Inside the column is the important component: the stationary phase composed of the high-boiling liquid. The liquid is usually impregnated on a high...


GOOD
Needs sources. (these aren't properly formatted) use APA or CHICAGO or MLA.
Concise. BUT Plagiarism is obviously present. Aren't you gonna credit people???? hmmm??
0 out of 0 people found this comment useful.