The ozone layer is a region of concentration of the ozone (O3) molecule in the Earth's atmosphere. The layer sits at an altitude of about 10 to 50 kilometers, with a maximum concentration in the stratosphere at an altitude of approximately 25 kilometers. In recent years, scientists have measured a seasonal thinning of the ozone layer primarily at the South Pole. This phenomenon is being called the ozone hole.
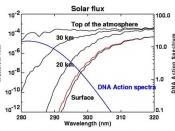
The ozone layer naturally shields Earth's life from the harmful effects of the sun's ultraviolet (UV) radiation. A severe decrease in the concentration of ozone in the ozone layer could lead to the effects of an increase in the incidence of skin cancer (UV radiation can destroy acids in DNA), a large increase in cataracts and sun burning, suppression of immune systems in organism, adverse impact on crops and animals, reduction in the growth of phytoplankton found in the Earth's oceans, and cooling of the Earth's stratosphere and possibly some surface climate effect.
Ozone is created naturally in the stratosphere by the combining of atomic oxygen (O) with molecular oxygen (O2). This process is activated by sunlight. Ozone is destroyed naturally by the absorption of ultraviolet radiation, and by the collision of ozone with other atmospheric atoms and molecules.
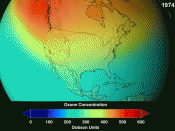
Since the late 1970s, scientists have discovered that stratospheric ozone amounts over Antarctica in springtime (September - November) have decreased by as much as 60 %. Satellite measurements (NIMBUS 7 - Total Ozone Mapping Spectrometer) have indicated a 3 % decrease in ozone between 65ð North - 65ð South since 1978. A reduction of about 3 % per year has been measured for Antarctica where most of the ozone loss is occurring globally. During the late 1990s, large losses of ozone were recorded above Antarctica year after year in the months...

Http://www.physicalgeography.net/fundamentals/7e.html
. . .
0 out of 0 people found this comment useful.